BIBA Publishing
Study finds significant cognitive impairment in adult survivors of childhood brain...
Cancer survivors of childhood brain tumours show significant deficits in intelligence, educational achievement and employment, even decades after treatment, an unprecedented study at St Jude Children's Research Hospital has found.
Bioness StimRouter neuromodulation system clinical trial results published
Bioness has announced the successful results from the company's one-year clinical trial of the StimRouter neuromodulation system, published in Neuromodulation.
Proton beam therapy may be as safe as radiotherapy for treatment...
The treatment was shown to have similar survival rates to conventional radiotherapy in a study published in The Lancet Oncology.
Natalizumab tied to rising John Cunningham virus levels in people with...
People taking natalizumab for the treatment of multiple sclerosis may be at a heightened risk of developing of progressive multifocal leukoencephalopathy.
Jan Medical secures US$7.5 million in series C funding from Brainlab
This funding will be used to complete ongoing clinical trials and filing of product de novo with the FDA, as well as funding CE mark registration in the EU, for BrainPulse.
First stroke patient treated with Rapid Medical’s Tigertriever
Using Rapid Medical's proprietary technology, this stent retriever is designed to be fully visible and controllable. The device can be adjusted by physicians to fit the dimensions of blood vessels causing acute ischaemic stroke, and is the first of its kind, according to the company.
Karolinska University Hospital to purchase Elekta’s Leksell Gamma Knife Icon
This intracranial stereotactic radiosurgery system is designed to offer precision radiosurgery to more patients with a wider variety of types and sizes of tumour.
First-line aspiration with Sofia device is “worth a try”
Thomas Liebig reported that a first-line aspiration approach with MicroVention's Sofia device is "worth a try" because it is associated with similar results to first-line aspiration approaches with other dedicated intermediate catheters, and unsuccessful aspiration with Sofia can be "easily converted" into stent retriever-based therapy.
Patient follow-up completed for BlueWind Medical’s Peripheral Neuropathic Pain study
The study evaluates the company's Reprieve wireless neurostimulation device, with a view to CE mark accreditation for multiple clinical indications.
Deep brain stimulation of the fornix (DBSf) shows benefit in the...
The phase 2 study appears to demonstrate acceptable safety and increased glucose metabolism in the treatment of patients with mild Alzheimer's disease.dbsf
NeuroPace RNS system receives ‘Most Promising New Product’ award
The closed-loop responsive neurostimulation system recieved the award at the 2015 Phoenix Medical Device CEO Conference
Seven US medical centres to acquire Elekta’s new Leksell Gamma Knife...
Seven centres are first in USA to order Icon, which features the first integrated stereotactic cone beam CT imaging, online adaptive dose evaluation and planning enabling the most accurate delivery of frameless and frame-based treatments, according to Elekta.
WEB .021″ approved for use in US IDE study
Sequent Medical has announced that the US Food and Drug Administration has approved the inclusion of WEB .021" devices for use in its US Investigational Device Exemption (IDE) clinical study called WEB-IT.
EmboTrap could be a “promising” device for acute ischaemic stroke
One of the newest devices to surface for the treatment of acute ischaemic stroke is the EmboTrap from Neuravi. At the World Live Neurovascular Course (WLNC; 8-10 June, Chicago, USA) Osama Zaidat and Tommy Andersson introduced the device and presented the results of the first 25 cases performed by Andersson and the team at Karolinska in Sweden.
New device is “equivalent to, or better than” known flow diverters...
In the first study to be published reporting on endovascular aneurysm treatment with the p64 flow modulation device (Phenox), Francesco Briganti and colleagues have found that the device provides a safe procedure with no technical complications.
AcuteCare Telemedicine partners with Emory Saint Joseph’s Hospital on Telestroke programme
Emory Saint Joseph's Hospital, Atlanta, USA, is improving treatment for stroke patients with the launch of a Telestroke programme, which allows neurologists to remotely evaluate patients around the clock through real-time conferencing with a Telerobot monitor.
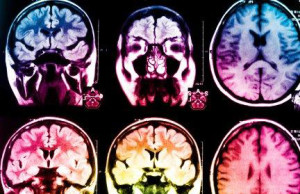
Alzheimer’s disease consists of three distinct subtypes according to UCLA study
The finding could lead to more highly targeted research and, eventually, new treatments for the debilitating neurological disorder, which robs people of their memories.
Researchers find evidence of link between stress and Alzheimer’s disease
The research, conducted on a mouse model and in human cells, found that a stress-coping hormone released by the brain boosts the production of protein fragments.
Alzheimer’s may affect the brain differently in African-Americans than European-Americans
The disease may cause different pathologies in African-Americans than in white Americans of European descent, according to a study published in an online issue of the medical journal of the American Academy of Neurology.
Three European health centres to use Elekta’s Leksell Gamma Knife Icon...
Three medical centres in the United Kingdom and Germany have ordered and are now planning upgrades to Elekta's latest generation stereotactic radiosurgery system for the brain, Leksell Gamma Knife Icon.
Southend University Hospital utilises 3D Systems’ simulator for life-saving team training...
Southend University Hospital established an innovative interventional stroke service using the ANGIO Mentor Suite simulator at Anglia Ruskin University.
In-home noninvasive brain stimulator may ease Parkinson’s symptoms
To give Parkinson's patients another in-home option, Johns Hopkins graduate students have invented a headband-shaped device to deliver noninvasive brain stimulation to help reduce the symptoms.
Strokes steal eight years’ worth of brain function, new study suggests
In both black and white patients, having had a stroke meant that their score on a 27-item test of memory and thinking speed had dropped as much as it would have if they had aged 7.9 years overnight.
Data demonstrates efficacy of Brainsway’s Deep TMS for treatment of ADHD...
New data demonstrating the efficacy of Brainsway's Deep TMS for the treatment of ADHD and OCD have been presented at the Society of Biological Psychiatry Annual Scientific Meeting ( 14-16 May, Toronto, Canada).
FDA approves Senza spinal cord stimulation system delivering HF10 therapy
Nevro has announced that it has received approval from the United States Food and Drug Administration (FDA) for its Senza spinal cord stimulation system....
Comprehensive stroke centres may improve bleeding stroke survival
People with haemorrhagic strokes are more likely to survive if they are treated at a comprehensive stroke centre, according to research published in the Journal of the American Heart Association.
Flowonix Medical announces first implants of Prometra II
Flowonix Medical has announced that the first implants of its Prometra II intrathecal infusion pump took place on 5 May, 2015 at St Francis Hospital in Charleston, USA.
First biosynthesised cellulose-based dural replacement for neurosurgery launched
DePuy Synthes CMF has announced the launch of Synthecel Dura Repair, the first commercially available biosynthesised dural replacement derived from cellulose for use in neurosurgery.
Cefaly to present breakthrough data in migraine treatment
On 14 May at a meeting of EUROHEADPAIN Cefaly Technology will present three significant patent pending systems of neuromodulation that are in development
Indications expanded for StealthStation electromagnetic surgical navigation technology
Medtronic plc has announced that it has received clearance from the US Food and Drug Administration (FDA) for expanded indications of specific StealthStation electromagnetic surgical navigation system instruments for paediatric and adult cranial and ENT procedures.
St Jude Medical completes acquisition of Spinal Modulation
Now complete, the acquisition of Spinal Modulation adds DRG stimulation technology to the St Jude Medical chronic pain portfolio.
Clinical trial moves stem cell therapy for ALS patients one step...
Stem cells are a safe therapy for patients with amyotrophic lateral sclerosis (ALS), according to the results of a recently completed phase 1 clinical trial.
Brain imaging can help differentiate between PTSD and traumatic brain injuries
By looking at the brain, scientists believe it is now possible to distinguish between two very different conditions that can have very similar symptoms.
Monteris Medical launches NeuroBlate SideFire Select and FullFire Select reduced diameter...
Monteris Medical has announced the launch of two new reduced diameter mini-probes for its NeuroBlate System, a minimally invasive robotic laser thermotherapy tool.
Codman Neuro launches Enterprise 2 in Europe
The Enterprise 2 system is the latest generation of the company's self-expanding stent and delivery system used to treat wide-necked intracranial aneurysms and to help maintain the position of endovascular coils.
Terrence L Cascino elected president of American Academy of Neurology
The American Academy of Neurology (AAN) has elected Terrence L Cascino, Mayo Clinic of Rochester, USA, as its 34th president.
Boston Scientific announces strategic collaboration with Brainlab
As part of the agreement, Boston Scientific will begin distributing the Brainlab deep brain stimulation surgical planning portfolio with the Boston Scientific Vercise deep brain stimulation system in select countries.
Medtronic announces consent decree with FDA for the SynchroMed drug infusion...
The company's efforts are focused on the implementation of design changes to the SynchroMed drug infusion pump to address issues the company has previously communicated, and on enhancing the Neuromodulation quality system.
New Leksell Gamma Knife to benefit more patients with brain disease
With the introduction of Elekta's new Leksell Gamma Knife Icon, the benefits of precision cranial radiosurgery are now available for more patients with a wider variety of tumour types and sizes.
Long-term exposure to air pollution may pose risk to brain structure,...
A new study led by scientists from Beth Israel Deaconess Medical Center and Boston University School of Medicine, USA, suggests that long-term exposure can cause damage to brain structures and impair cognitive function in middle-aged and older adults.
For lower-grade brain blood vessel malformations, surgery has “excellent clinical outcomes”
Interventional treatments-especially surgery-provide good functional outcomes and a high cure rate for patients with lower-grade arteriovenous malformations of the brain, reports the May issue of Neurosurgery.
Effect of Lemtrada on slowing brain atrophy and MRI lesion activity...
In relapsing remitting multiple sclerosis (RRMS) patients treated with Lemtrada in the phase III pivotal studies, MRI effects observed in the two-year trials were maintained through two additional years in the extension study (years three and four).
Medtronic announces European approval of the first and only full-body MR...
The expanded approval for full-body MRI scans applies to all patients receiving a new system and to an estimated 13,000 people in Europe already receiving Medtronic DBS therapy.
FDA clears MRI-compatible MEMS cannula
The Alcyone MEMS cannula is a dual-lumen, MRI-compatible injection and aspiration cannula for use in the brain.
Drugs stimulate body’s own stem cells to replace the brain cells...
A pair of topical medicines already alleviating skin conditions each may prove to have another, even more compelling use: instructing stem cells in the brain to reverse damage caused by multiple sclerosis.
Clinical phase II study with NeuroSTAT for traumatic brain injury passes...
The study of NeuroVive's drug candidate NeuroSTAT will continue as planned and move on to the next dosage group following endorsement from the independent safety committee.
NurOwn demonstrates a statistically significant effect in ALS patients
BrainStorm Cell Therapeutics presented results from its phase 2a study of NurOwn in amyotrophic lateral sclerosis (ALS) at a poster session at the American Academy of Neurology annual meeting, in Washington, DC, USA.
St Jude Medical announces intent to acquire Spinal Modulation
Following the completion of this acquisition, St Jude Medical will become the only medical device manufacturer to offer radiofrequency ablation (RFA), spinal cord stimulation (SCS) and dorsal root ganglion (DRG) stimulation therapy solutions for the treatment of chronic pain.
Two different carotid artery stenting procedures show little difference in effectiveness
Use of either proximal embolic protection devices or distal filter embolic protection devices during elective carotid artery stenting results in low rates of in-hospital stroke and death, according to a new study.
Evidence builds for endovascular treatment of acute ischaemic stroke
With the publication of two more stroke trials, the evidence in favour of endovascular treatment in patients with acute ischaemic stroke has reached new heights. Data from SWIFT PRIME and REVASCAT add to that of three other trials-MR CLEAN, EXTEND-IA and ESCAPE-that have begun to change the face of ischaemic stroke treatment.
First US commercial procedure with the Enroute transcarotid neuroprotection system
Silk Road Medical has announced the first US commercial procedure using the Enroute transcarotid neuroprotection system was successfully performed at Mills-Peninsula Medical Center in Burlingame, USA by vascular surgeon John E Rosenman.
New research study to demonstrate value of PET scans in Alzheimer’s...
The IDEAS study will assess the impact of brain amyloid PET imaging on a variety of patient outcomes.
Clinical utility of real-time navigated laser therapy for lesion ablation within...
Three prominent neurosurgeons will review their clinical experience using real-time navigated laser therapy for brain lesion ablation within intraoperative MRI suites at the upcoming 83rd American Association of Neurological Surgeons (AANS) Annual Scientific Meeting.
The ALS Association to collaborate with GlaxoSmithKline on new ALS clinical...
The ALS Association, Harvard Stem Cell Institute, and Massachusetts General Hospital Neurological Clinical Research Institute have announced that they are collaborating with GlaxoSmithKline on a clinical trial to evaluate the potential of an anti-epileptic drug in ALS patients.
Women and men have different exclusion criteria for clot-busting stroke drug,...
After analysing stroke treatment records, researchers at Rhode Island Hospital in collaboration with researchers from the University of Cincinnati, both USA, learned that women and men have different reasons for being excluded from receiving the common clot-dissolving drug, recombinant tissue plasminogen activator (rt-PA).
Jan Medical’s Nautilus BrainPulse detected cerebral vasospasm with clinically meaningful accuracy...
A clinical study published in Neurocritical Care demonstrated that Nautilus BrainPulse is a highly sensitive skull accelerometry that can detect cerebral vasospasm "with clinically meaningful accuracy".
Drug restores brain function and memory in early Alzheimer’s disease
A novel therapeutic approach for an existing drug reverses a condition in elderly patients who are at high risk for dementia due to Alzheimer's disease, researchers at Johns Hopkins University found.
Stimwave Technologies announces partnership with Epimed International
The partnership, in addition to co-product development, will support distribution of Stimwave's Wireless Pain Relief technology, which became available in the USA in January 2015.
Researchers build brain-machine interface to control prosthetic hand
New University of Houston research has demonstrated that an amputee can grasp with a bionic hand, powered only by his thoughts.
Stimwave appoints Gabor Racz as chair of Medical Advisory Board
Racz has been honoured with several lifetime achievement awards from societies throughout the world in recognition of contributions that span more than five decades.
AANS, CNS and Joint Cerebrovascular Section endorse interventional thrombectomy in treatment...
The three groups endorse the approach in their article, "MR CLEAN: past the tipping point of clinical equipoise," by Henry H Woo et al, published online, ahead of print, in the Journal of Neurosurgery.
High-frequency surpasses traditional spinal cord stimulation in first controlled trial comparing...
The first-ever randomised, controlled trial to compare spinal cord stimulation technologies found that high-frequency stimulation using 10kHz (HF10) exceeded lower-frequency, traditional stimulation in response rate and pain relief.
Health Canada greenlights IMRIS ceiling-mounted intraoperative CT solution
IMRIS Inc has announced that VISIUS iCT, the first and only ceiling-mounted intraoperative computed tomography scanner, has received Health Canada licensing allowing for sales and marketing in the country.
Use of stent, compared to medications, increases risk of stroke in...
The VISSIT study has shown that among patients with symptomatic intracranial arterial stenosis, the use of a balloon-expandable stent compared with medical therapy (clopidogrel and aspirin) resulted in an increase of stroke or transient ischaemic attack.
Brain tumour cells decimated by mitochondrial “smart bomb”
An experimental drug that attacks brain tumour tissue by crippling the cells' energy source called the mitochondria has passed early tests in animal models and human tissue cultures, say Houston Methodist scientists.
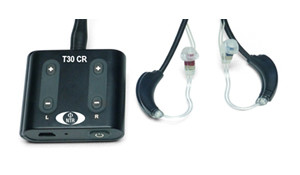
New tinnitus treatment study reduces total symptoms by almost 40%
A year-long study shows that Acoustic CR neuromodulation treatment is effective in reducing tonal tinnitus symptoms such as severity and loudness and annoyance by nearly 40%.
Troops who do not pass the smell test are likely to...
Decreased ability to identify specific odours can predict abnormal neuroimaging results in blast-injured troops, according to a new study by US federal researchers released online in the journal Neurology.
UCSF team finds key to making neurons from stem cells
A research team at UC San Francisco has discovered an RNA molecule called Pnky that can be manipulated to increase the production of neurons from neural stem cells.
More neurosurgeons will be required to handle increased brain bleeds
By 2030, chronic subdural haemorrhage will be the most common adult brain condition requiring neurosurgical intervention in the USA, and hospitals and neurosurgeons may be under-manned to handle the projected onslaught of patients, a new study says.
Is it dementia, or just normal aging? New tool may help...
Researchers at Mayo Clinic developed a new scoring system to help determine which elderly people may be at a higher risk of developing the memory and thinking problems that can lead to dementia.
John Theurer Cancer Center joins study exploring novel delivery of cancer-selective...
In this study, Toca 511 is injected directly into the brain tumour through a stereotactic biopsy needle. Once injected, Toca 511 infects tumour cells, sparing healthy functioning brain cells.
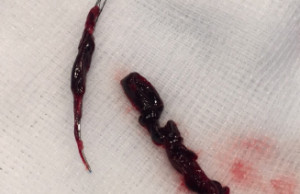
Cover device proves more effective than guide catheter in stroke model
In an in vitro stroke model comparison, the use of the Cover accessory device (Lazarus Effect) in conjunction with a stent retriever resulted in higher successful recanalisation rates, no embolic events and proved to be more effective than the use of a conventional guide catheter or a balloon guide catheter.
Feinstein Institute researcher to receive Potamkin Prize for Alzheimer’s research
The Potamkin Prize honours researchers for their work in helping to advance the understanding of Pick's disease, Alzheimer's disease and related disorders.
New technique could lead to long-lasting localised stimulation of brain tissue...
Researchers at MIT have developed a method to stimulate brain tissue using external magnetic fields and injected magnetic nanoparticles-a technique allowing direct stimulation of neurons, which could be an effective treatment for a variety of neurological diseases, without the need for implants or external connections.
Neuralstem announces topline results of phase II ALS trial
Neuralstem has announced top line data from the phase II trial of NSI-566 spinal cord-derived neural stem cells under development for the treatment of amyotrophic lateral sclerosis (ALS).
Using robots for stroke rehabilitation
Researchers have developed a prototype of a robotic glove which stroke sufferers can use in their own home to support rehabilitation and personal independence in receiving therapies.
electroCore non-invasive vagus nerve stimulation is effective in treating cluster headaches
Seventy-nine per cent of patients who completed the trial (15 out of 19) reported an overall improvement in their condition.
One-third of Americans do not have access to stroke centre within...
Even under optimal conditions, a large proportion of the population would be unable to access a stroke centre within this window, according to a new study.
Mouth guards gather new information that may lead to more accurate...
The mouth guards were able to measure six degrees of freedom, including rotational movement (roll, pitch, and yaw), while current safety standards use three degrees of freedom.
Direct brain neurostimulation for partial onset seizures provides long-term benefit
A study of a direct brain responsive neurostimulator found that responsive direct cortical stimulation reduces seizures and improves quality of life over an average of 5.4 years for for partial onset seizures that cannot be controlled with medication
Next generation Medina coil system shows improvements in early human experience
In early use of the Medina coil system, researchers have found the device to be a next generation coil that combines the familiar procedural safety and technique associated with conventional coils, with improved circumferential aneurysm filling, which, they say, it is thought will lead to improved long-term outcomes.
Findings may help with the management of anticoagulant-related bleeding within the...
Reversal of international normalised ratio below a certain level and systolic blood pressure less than 160mmHg at four hours were associated with lower rates of haemorrhage enlargement in oral anticoagulation-associated intracerebral haemorrhage patients, according to a new study.
Long sleepers may have an increased risk of stroke
A new study has found that people who slept more than eight hours a night were 46% more likely to have a stroke than people who slept six to eight hours a night.
FDA clears Bioness StimRouter neuromodulation system
The StimRouter is a minimally invasive implantable device designed to reduce pain by specifically targeting the affected peripheral nerve.
Medtronic acquires Advanced Uro-Solutions
Advanced Uro-Solutions develops and manufactures the NURO percutaneous tibial nerve stimulation system for patients with overactive bladder and associated symptoms.
Resistance to aspirin tied to more severe strokes
Patients who were aspirin resistant also had larger areas of the brain affected by the stroke, as measured by MRI diffusion weighted imaging.
SANTE study shows DBS therapy for treatment-resistant epilepsy demonstrates significant and...
Medtronic DBS therapy for epilepsy was associated with a sustained and statistically significant reduction in seizure frequency from baseline that continued to improve over time: 69% median seizure reduction from baseline at five years and 41% at one year.
Saluda Medical secures US$10 million in new financing
Saluda Medical has announced that it has received US$10 million in Series B financing bringing a breakthrough treatment for chronic pain one step closer to reality.
Development of personalised cellular therapy for brain cancer
Preclinical study results pave the way for newly-opened clinical trial of immune cells engineered to attack protein found on tumours in 30% of patients with glioblastoma.
New device improves healing of some ruptured aneurysms
Research presented at the American Stroke Association's International Stroke Conference (Nashville, USA, 11-13 February, 2015) suggests that a new device inserted into small ruptured brain aneurysms significantly improved healing of ruptured aneurysms compared to a standard device.
Blood flow technology from VasSol Identifies risk of recurrent stroke
The results, presented at the International Stroke Conference (Nashville, USA, 11-13 February, 2015) are expected to substantially improve both the study and treatment of stroke.
Caputron Medical signs distribution agreement with Neurosoft
The distribution agreement gives Caputron Medical the rights to market the Neuro-MS/D Diagnostic, Therapeutic, and Advanced Therapeutic rTMS stimulators in the USA and Canada.
American Stroke Association and Medtronic collaborate to reduce recurrent strokes
The two organisations will work together over several years to educate, raise awareness and support effective management of patients who have strokes.
Stem cell transplants may work better than existing drug for severe...
Those who received the stem cell transplants had 80% fewer new areas of T2 lesions than those who received mitoxantrone.
US stroke patients receiving better and more timely care
More ischaemic stroke patients are now being quickly transferred to certified US stroke centres, according to research presented at the American Stroke Association's International Stroke Conference 2015.
Mobile stroke units improve response times and outcomes for patients
There was a significant reduction of median alarm to CT scan completion times-41 minutes in MSTU vs. 62 minutes in emergency room patients.
Groundbreaking studies find that neurointerventional surgery reduces stroke mortality
Two new clinical trials on the treatment of stroke (ESCAPE and EXTEND IA) demonstrate that neurointerventional surgery significantly increases the number of patients who are able to live independently without major neurological disabilities.
Creatine does not slow rate of Parkinson’s disease progression
Treatment with creatine monohydrate for at least five years for patients with early and treated Parkinson's disease failed to slow clinical progression of the disease, compared with placebo, according to a study in JAMA.
Human stem cells repair damage caused by radiation therapy for brain...
Investigators used lab-grown oligodentrocyte progenitor cells to treat rats that had been exposed to brain irradiation.
FDA clears Enroute transcarotid neuroprotection system
The Enroute system (Silk Road Medical) is a first in class system used to directly access the common carotid artery and initiate high rate temporary blood flow reversal to protect the brain from stroke while performing carotid angioplasty and stenting.
Trial results show Brainsway deep TMS to be efficacious and safe...
A multicentre double blinded randomised controlled trial has shown Brainsway deep TMS (transcranial magnetic stimulation) to be efficacious and safe for drug-resistant depression, with a stable effect over three months of maintenance treatment.
FDA approves Pipeline Flex embolisation device
Designed to divert blood flow away from an aneurysm, the Pipeline Flex embolisation device (Medtronic) features a braided cylindrical mesh tube that is implanted across the base or neck of the aneurysm.
Transradial access valid for treatment of acute ischaemic stroke
According to a recent retrospective review published in the Journal of NeuroInterventional Surgery, the transradial approach to mechanical embolectomy is a valid approach for the endovascular treatment of acute ischaemic stroke.
NeuroPace RNS system reduces seizures and improves quality of life for...
Long-term treatment study data show sustained effectiveness in adults historically resistant to other treatments.
BrainStorm announces positive DSMB recommendation in ongoing NurOwn amyotrophic lateral sclerosis...
The DSMB did not find any lab abnormalities, adverse events or significant protocol deviations that would be cause for concern.
Heavy drinking in middle-age may increase stroke risk more than traditional...
In a study of 11,644 middle-aged Swedish twins who were followed for 43 years, researchers compared the effects of an average of more than two drinks daily to less than half a drink daily.
Low-frequency deep brain stimulation improves difficult-to-treat Parkinson’s symptoms
60Hz stimulation more effective than standard treatment at improving swallowing and gait issues
Review confirms that electroCore’s non-invasive vagus nerve stimulation is safe and...
The review examined the efficacy of both surgically implanted non-invasive vagus nerve stimulation, including electroCore's gammaCore device.
Brain imaging may help predict future behaviour
Brain imaging can help predict an individual’s future learning, criminality, health-related behaviours, and response to drug or behavioural treatments.
Stem cell transplantation shows potential for reducing disability in multiple sclerosis...
Treatment with nonmyeloablative hematopoietic stem cell transplantation was associated with improvement in measures of disability and quality of life
Siemens Healthcare and IMRIS bring hybrid ORs to Sahlgrenska University Hospital...
This sale involves both radiology and MRI systems in four hybrid operating rooms for the hospital’s new imaging centre with clinical opening planned in 2016.
Link discovered between protective effects of cooling on the brain and...
Researchers have identified a protective mechanism that activates when body temperature is lowered, initiating a process that prevents the loss of brain cells and the connections between them.
InVivo Therapeutics announces reopening of enrolment for ongoing pilot trial
This is the company's first clinical study of its investigational degradable polymer Neuro-Spinal Scaffold to treat patients with acute spinal cord injury.
Ischaemic micro-lesions associated with flow-diverting stents in treatment of intracranial aneurysms
Researchers from Dupuytren University Hospital, Limoges, France, conducted a study to determine the extent of these complications and their implications on clinical outcomes in flow-diverting stent treatments.
Non-invasive EEG reveals depolarisations
New research at the University of Cincinnati Neuroscience Institute has shown that spreading depolarisations can be measured by the placement of electroencephalograph (EEG) electrodes on the scalp. Head of the research team, Jed Hartings speaks to NeuroNews about the discovery and its potential to change current practice.
PulseRider safe and effective in early USA experience
Initial experience with PulseRider (Pulsar Vascular) has shown the device to be safe and effective as an adjunct in the treatment of bifurcation aneurysms arising at the basilar apex or carotid terminus.
Publication demonstrates high accuracy for identification of acute stroke
BrainScope has announced the publication of an independent study that demonstrated the potential clinical utility of its traumatic brain injury technology to identify acute stroke in the hospital Emergency Department setting.
Update regarding coverage of VNS Therapy system in treatment-resistant depression
Cyberonics has announced the receipt of a decision from the Departmental Appeals Board (DAB) of the Department of Health and Human Services in the USA.
Stem cell transplants for Parkinson’s disease edging closer
A major breakthrough in the development of stem cell-derived brain cells has put researchers on a firm path towards the first ever stem cell transplantations in people with Parkinson's disease.
Can exercise help people with Parkinson’s disease?
Exercise may help people with Parkinson's disease improve their balance, ability to move around and quality of life, even if it does not reduce their risk of falling, according to a new study published in the online issue of Neurology.
Stroke falls to No. 5 cause of death in USA
Stroke has dropped from the USA's fourth-leading cause of death to No. 5, according to new federal statistics.
Stem cell transplants may halt progression of multiple sclerosis
Three-year outcomes suggest that high-dose immunosuppressive therapy followed by transplantation of a person's own blood-forming stem cells may induce sustained remission in some people with relapsing-remitting multiple sclerosis.
NeuroMetrix to showcase Quell wearable pain relief technology
NeuroMetrix will unveil and demonstrate Quell, a novel wearable pain relief device at the 2015 International Consumer Electronics show.
Seventy-one teams to compete in NIH Neuro Startup Challenge
The Challenge has teams competing to commercialise 16 National Institutes of Health-conceived and -developed inventions involving therapeutics, diagnostics, prognostics, and medical devices for a range of brain diseases.
Alcyone Lifesciences appoints Michael Rogawski to Clinical and Scientific Advisory Board
Rogawski is an internationally recognised expert on treatments for seizures and epilepsy.
Longer cooling, lower temperature no improvement for infant oxygen deprivation
The standard treatment for newborns whose brains were deprived of oxygen appears to work better than proposed alternatives, according to a study from a National Institutes of Health research network.
CHMP recommends approval of Xadago to treat Parkinson’s disease in the...
The EU Committee for Medicinal Products for Human Use (CHMP) recommended that the European Commission approve the use of Xadago (safinamide) as add-on to L-dopa alone or in combination with dopamine agonists, entacapone, amantadine, and/or anticholinergics, for the treatment of patients with mid-late stage Parkinson’s disease experiencing motor fluctuations despite being stabilised on 'Standard of Care'.
MR CLEAN: Better outcomes with intervention in ischaemic stroke patients
For the first time, the Multicentre randomised clinical trial of endovascular treatment for acute ischaemic stroke in the Netherlands (MR CLEAN) has shown better outcomes in favour of intervention in patients with acute ischaemic stroke.
American Academy of Neurology calls for more research on medical marijuana...
The AAN supports the reclassification of marijuana-based products by the federal government to improve access for study of marijuana or cannabinoids under approved research protocols.
Brainlab multiple metastases planning software available in the USA
Adaptive Hybrid Surgery and Automatic Brain Metastases Planning software by Brainlab are now available in the USA after successful clinical use in several international markets.
Stroke treatment in England varies widely by location
The figures released by the HSCIC show that the percentage of patients admitted to an acute stroke unit within four hours of arrival at hospital varied from 22% to 85% across different clinical commissioning groups.
Cyberonics reports positive results from AspireSR generator vagus nerve stimulation studies
The E-36 and E-37 studies assessed the acute impact of the AspireSR generator on seizure duration and termination, as well as the long-term evaluation of safety, clinical benefit of the automatic stimulation feature, and quality of life.
Brainsway receives IDE approval from FDA for launching pivotal PTSD multicentre...
Brainsway has announced that it has received an IDE (Investigational Device Exemptions) approval from the FDA for launching a multicentre trial which will examine the effectiveness of Brainsway's Deep TMS (deep transcranial magnetic stimulation) for treating PTSD (post-traumatic stress disorder) patients.
Research shows promise for the DRG as a target for neuromodulation
At the 18th annual North American Neuromodulation Society (NANS) congress, researchers presented 20 international, peer-reviewed scientific abstracts from Australia and Europe highlighting clinical results of the Axium neurostimulator system for the treatment of chronic, intractable pain.
Memory lapses among highly educated may signal higher stroke risk
People with a high level of education who complain about memory lapses have a higher risk for stroke, according to new research in the American Heart Association journal Stroke.
Acorda initiates phase 3 trial of CVT-301 in Parkinson’s disease
Acorda Therapeutics has enrolled its first patient in a phase 3 study of CVT-301 for the treatment of "off" episodes in Parkinson's disease.
Images of brain after mild stroke predict future risk
A CT scan of the brain within 24 hours of a mild, non-disabling stroke can predict when patients will be at the highest risk of another stroke or when symptoms may worsen.
First US patients receive Genzyme’s Lemtrada following FDA approval
Genzyme has announced that the first US patients have initiated treatment with Lemtrada in the commercial setting following its FDA approval for the treatment of patients with relapsing forms of multiple sclerosis.
Brainsway receives CE approvals for treating MS, stroke and obsessive compulsive...
Brainsway has announced that it has received a CE marketing approval, enabling it to immediately market and sell Brainsway deep TMS (deep transcranial magnetic stimulation) in Europe, for treatment of multiple sclerosis (MS), stroke and obsessive compulsive disorder (OCD).
Spinal Modulation completes enrolment of US pivotal trial evaluating the Axium...
The Axium system is a targeted form of spinal cord stimulation (SCS), which uses an implantable medical device to deliver mild electrical pulses that mask or interrupt pain signals as they travel from the periphery to the brain.
IMRIS completes SYMBIS surgical system human factors study
IMRIS has successfully completed the human factors study for the SYMBIS surgical system - the validation used to support the product's 510(k) submission to the US Food and Drug Administration.
Eli Lilly and Company and AstraZeneca initiate early Alzheimer’s disease pivotal...
Eli Lilly and Company and AstraZeneca have announced enrolment of the first patient into AMARANTH, a phase II/III study of an oral beta secretase cleaving enzyme inhibitor currently in development as a potential treatment for Alzheimer's disease.
Same regions in brain are vulnerable to both Alzheimer’s and schizophrenia
Researchers have found a specific network of brain regions that is not only more vulnerable to unhealthy ageing, for example, Alzheimer's disease, but also to disorders that emerge in young people, such as schizophrenia.
InVivo Therapeutics announces OHSU as fifth clinical trial site for Neuro-Spinal...
InVivo Therapeutics has announced that the Oregon Health & Science University (OHSU) in Portland, Oregon, USA, is the fifth clinical site in the company's ongoing IDE pilot study of its Neuro-Spinal Scaffold in patients with acute spinal cord injury (SCI).
Repetitive transcranial magnetic stimulation studied for stroke rehabilitation
Researchers at The Ohio State University Wexner Medical Center are attempting to improve arm movement in stroke patients by using a transcranial magnetic stimulator (TMS) device.
New Alzheimer’s programme to focus on prevention, intervention, research and support
Cedars-Sinai has launched the Alzheimer's Prevention Program to help identify patients at risk of developing the neurological disorder and to reduce the impact on those diagnosed with the condition.
Barrow neurosurgeons implant the world’s first scaffold into a patient’s spinal...
Performed last month, the surgery involves inserting a bioresorbable scaffolding implant to act as a bridge across the gap of the injured section of the cord in an attempt to help the spinal cord heal.
Portable electric device found to slow and reverse growth of glioblastoma
A non-invasive, portable electrical device has been found to lengthen the lives of some patients suffering from glioblastoma, the most common and deadliest form of brain cancer.
FDA clears Ahead 100 device for adjunctive assessment of traumatic brain...
The Ahead 100 from BrainScope uses a patient's electroencephalograph (EEG) to provide an interpretation of the structural condition of the patient's brain after head injury.
MR CLEAN: expert opinion
Since the first presentation of data at the World Stroke Congress, the MR CLEAN trial has sent a ripple across the entire neurointerventional arena, raising questions about the future of stroke treatment and the fate of similar trials, some of which have already been halted.
Majority of UK neurosurgical units following published recommendations, but room for...
It has been discovered via a UK national survey of current practice that the majority of neurosurgical units have been following most of the key recommendations for the management of aneurysmal subarachnoid haemorrhage.
CE mark for new Asahi Intecc neurovascular guide wires
Asahi Intecc has received CE certification of neurovascular guide wires: Asahi Chikai 008, Asahi Chikai black, and Asahi Chikai black 18 and is initiating sales in Europe.
Lazarus Effect Cover device receives CE mark
The Lazarus Cover is a nitinol-mesh cover that surrounds a retriever device and captures material during removal from a blood vessel.
Studies support the use of the CyberKnife system as a safe...
Accuray Incorporated has announced that studies presented at the 15th European Congress of Neurosurgical Societies (EANS) in Prague, October 12 - 17, 2014 reinforce the benefits of the CyberKnife robotic radiosurgery system for the treatment of neurological diseases such as meningiomas, acoustic neuromas, trigeminal neuralgia, and spinal tumours.
NeuroSigma receives notice of allowance for subcutaneous trigeminal nerve stimulation patent
The patent application is related to the use of trigeminal nerve stimulation with minimally invasive, subcutaneously implanted electrodes and pulse generator for the treatment of a wide variety of medical disorders.
ALS Association triples research spending following ice bucket donations
The ALS Association Board of Trustees has developed a long-term strategy for leveraging the Ice Bucket Challenge donations it has received to advance its integrated mission of leading the fight to treat and cure ALS through global research, care services and public policy.
American Shared Hospital Services announces 2015 CMS gamma knife and proton...
American Shared Hospital Services has announced that the Centers for Medicare and Medicaid Services has posted its final Medicare hospital outpatient prospective payment rates for calendar year 2015. The rates for gamma knife and proton therapy remained as proposed earlier this year.
Over US$30m in funding allocated to rare brain disease research
Researchers studying frontotemporal degeneration disease, a leading cause of early onset dementia, will receive more than US$30m over the next five years in grants from the National Institutes of Health (NIH).
Diets high in fruit, vegetables, whole grains and nuts lower first-time...
Eating Mediterranean or DASH-style diets, regularly engaging in physical activity and keeping blood pressure under control can lower a person's risk of a first-time stroke.
Researchers join Biogen Idec to advance drug discovery for neurodegenerative diseases
Two leading neuroscientists have joined Biogen Idec to advance its research in neurodegenerative diseases.
New test to help brain injury victims recover
A dynamic new assessment for helping victims of trauma to the brain, including those suffering from progressive conditions such as dementia, has been developed by a clinical neuropsychologist at the University of Leicester, UK.
Traumatic brain injury associated with increased dementia risk in older adults
Traumatic brain injury appears to be associated with an increased risk of dementia in adults 55 years and older, according to a study published online by JAMA Neurology.
Encouraging trial results for electroCore’s non-invasive vagus nerve stimulation treatment for...
In an open label trial, electroCore's handheld non-invasive vagus nerve stimulation device gammaCore, completely resolved 44.8% of migraines within 30 minutes, with an additional 11.4% experiencing moderate benefits (incomplete resolution of their headaches) by two hours.
The Roskamp Institute discovers new target for drugs to treat Alzheimer’s...
Scientists at the Roskamp Institute have isolated a key molecule that gives researchers a new drug target for the treatment of the progressive, irreversible neurological disorder. This finding is the culmination of more than 10 years of work by more than a dozen scientists and clinicians in the research team in Sarasota.
Thymosin beta-4 crosses blood-brain barrier in animal stroke model
Researchers from RegeneRx Biopharmaceuticals have reported on study findings in which thymosin beta-4 crossed the blood-brain barrier in an animal stroke model.
Costs to treat bleeding strokes increases 10 years later
Costs to treat strokes caused by bleeding in the brain may increase significantly 10 years later, according to a study in the American Heart Association journal Stroke.
ALS Association and NEALS issue request for proposals for phase II...
ALS ACT, the ALS Association, and NEALS clinical trials have announced a call for phase II clinical trial applications for novel, high-potential treatments in amyotrophic lateral sclerosis (ALS).
New NICE guidance to tackle inequalities in multiple sclerosis care
People with multiple sclerosis should be offered a rapid and accurate diagnosis of their condition and access to specialist advice and proven therapies, says the National Institute for Healthcare and Excellence.
A new window of opportunity to prevent cardiovascular and cerebrovascular diseases
Mount Sinai experts show neuroimaging tests can reveal disruptive brain changes resulting from vascular disease risk factors before the appearance of any signs or symptoms of cardiovascular or cerebrovascular disease.
Codman Neuro launches first disposable forceps with dual irrigation
Codman Neuro has launched the Spetzler Malis dual irrigating disposable non-stick bipolar forceps, the first disposable dual irrigating forceps for neurosurgery.
Stenting safe and effective for long-term stroke prevention
Using stents to keep neck arteries open is just as effective as invasive neck surgery for long-term prevention of fatal and disabling strokes, reports an international trial led by University College London.
AVP-923 phase II results in Alzheimer’s patients presented at ANA meeting
Results from the phase II study evaluating AVP-923 for the treatment of agitation in patients with Alzheimer's disease were presented at the 2014 American Neurological Association meeting.
FDA and CE mark approval for new CoverEdge surgical leads
Boston Scientific has received FDA and CE mark approval for the CoverEdge 32 and CoverEdge X 32 surgical leads, designed for use with the Precision Spectra spinal cord stimulator system.
Delay the Disease Parkinson’s programme gets participants moving again
OhioHealth "Delay the Disease" is designed to empower those living with Parkinson's disease by optimising their physical function and helping to delay the progression of symptoms associated with the disease.
First patient enrolled in pilot spinal cord injury trial
The objective of the pilot study is to evaluate the safety and feasibility of the Neuro-Spinal Scaffold (InVivo Therapeutics) as well as to gather preliminary evidence of effectiveness.
NeuroSigma partners with US Veterans Administration for eTNS trial
NeuroSigma has has entered into a Cooperative Research and Development Agreement with the US Veterans Affairs for a phase I clinical trial to evaluate the benefits of non-invasive, external trigeminal nerve stimulation (eTNS) for patients with traumatic brain injury.
Drug-eluting balloon angioplasty shows excellent results for refractory recurrent carotid in-stent...
A long-term follow-up study reports on the safety and success of drug-eluting balloon angioplasty in treating patients with carotid in-stent restenosis that has proven refractory to treatment with balloon angioplasty and/or redo stenting.
Disputed theory on Parkinson’s origin strengthened
Researchers at Lund University, Sweden, have mapped the spread of Parkinson's disease from the gut to the brain in rat models.
Treeway BV and Leiden Academic Center for Drug Research collaborate on...
Treeway and Leiden Academic Center for Drug Research (LACDR) at the University of Leiden join forces and form a collaboration focused on the optimisation of clinical trial designs and data-analysis for ALS through the use of population disease progression models.
Neuravi receives US patents for neurovascular clot capture and retrieval
Neuravi has announced that the US Patent Office has approved two patents covering its therapeutic platform for the endovascular treatment of acute ischaemic stroke.
Mechanism that repairs brain after stroke discovered
A previously unknown mechanism through which the brain produces new nerve cells after a stroke has been discovered at Lund University and Karolinska Institutet in Sweden.
New Envoy catheters launched
Codman Neuro has announced the Europe, Middle East and Africa launch of the Envoy DA XB Distal Access Guiding Catheter and the 7F Envoy Guiding Catheter for neurovascular procedures.
Neuroscientists claim rare pair of research grants
In a rare distinction for one university, neuroimaging world leaders and University of Southern California (USC) Professors Arthur Toga and Paul Thompson will receive two major research centre awards to advance their exploration of the human brain.
IMRIS intraoperative imaging solutions to be featured at CNS
IMRIS has announced that use of intraoperative imaging for optimising workflow with neurosurgical focal therapies will be featured in its exhibition space at the Congress of Neurosurgical Surgeons (CNS) meeting (20-22 October, Boston, USA).
First patient treated in US pivotal trial evaluating cerebral protection during...
The randomised controlled SENTINEL Trial is the first in the USA to study capture and removal of debris released during TAVI that may otherwise be the source of stroke.
Kadimastem approached the FDA regarding its ALS treatment
Israeli biotechnology company Kadimastem has announced that it has approached the FDA regarding the cellular treatment it is developing for ALS. In the framework of talks with the FDA, Kadimastem intends to consolidate a preliminary outline for its continuing trials for this indication.
ALS Association announces initial commitment of US$21.7 million from Ice Bucket...
The ALS Association has approved an initial expenditure of US$21.7 million in funding to support six programmes and initiatives to expedite the search for treatments and a cure for amyotrophic lateral sclerosis (ALS). Additionally, US$12.5 million in matching donations bring the total commitment to US$34.2 million.
IMRIS horseshoe headrest named Life Sciences Alley New Technology Showcase winner
IMRIS announced that its horseshoe headrest has been selected one of 10 New Technology Showcase Winners by Twin Cities-based Life Sciences Alley (LSA), the USA's largest regional medical industry association.
Collaboration established to advance diagnostic candidate to detect CTE in former...
Aethlon Medical and its diagnostic subsidiary, Exosome Sciences, have announced that a clinical collaboration with the Boston University CTE Center has been established to advance a blood-based diagnostic candidate that could identify Chronic Traumatic Encephalopathy (CTE) in living individuals.
Medina Medical announces CE mark for its embolisation coil
Medina Medical has announced that it has received CE mark authorisation for its Embolization Framing Coil for commercial distribution in the European Union.
Neuralstem ALS investigator presents long-term follow-up phase I data
Neuralstem has announced that Jonathan D Glass presented long-term follow up data on the phase I trial testing NSI-566 human neural stem cells in the treatment of amyotrophic lateral sclerosis (ALS).
Randomised data show gammaCore significantly reduces cluster headache attack frequency
A large scale randomised clinical trial of the gammaCore therapy in chronic cluster headache not only significantly reduced the number of cluster headache attacks beyond the best available standard of care, but patients also experienced a greater reduction in number of attacks the longer they stayed on treatment.
EBS expands commercialisation of Next Wave non-invasive electrical brain stimulation device
The EBS Next Wave brain stimulation device is designed to expand the visual field of patients with impaired vision caused by glaucoma, stroke and other neurological diseases.
Neuroimaging technique identifies concussion-related brain disease in living brain
An experimental positron emission tomography (PET) tracer is effective in diagnosing concussion-related brain disease while a person is still alive, according to a case study.
Nexstim plans an Initial Public Offering on NASDAQ OMX First North
Nexstim has announced its intention to proceed with an Initial Public Offering of Nexstim on NASDAQ OMX First North Finland and NASDAQ OMX First North Sweden ("Admission"). It is expected that the Admission will occur in the 4th Quarter of 2014.
ATL1102 phase II trial results published in Neurology
Antisense Therapeutics has reported the publication of previously generated phase IIa clinical trial data on ATL1102 in the medical journal Neurology.
Evidence supports deep brain stimulation for obsessive-compulsive disorder
Available research evidence supports the use of deep brain stimulation for patients with obsessive-compulsive disorder who do not respond to other treatments, concludes a review in the October issue of Neurosurgery.
Mayo researchers reveal pathway that contributes to Alzheimer’s disease
Researchers have discovered a defect in a key cell-signalling pathway they say contributes to both overproduction of toxic protein in the brains of Alzheimer's disease patients as well as loss of communication between neurons - both significant contributors to this type of dementia.
Multiple sclerosis drug candidate shows new promise
Positive new data have been released on a drug candidate for relapsing multiple sclerosis that was first discovered and synthesised at The Scripps Research Institute.
CE mark for Vercise deep brain stimulation system for patients with...
Boston Scientific has received CE mark approval for the Vercise deep brain stimulation system for the treatment of tremor, including the most common form of this movement disorder known as essential tremor.
New mobile apps support education, safety, and adherence needs of seizure...
Adherent Health has announced the introduction of patient support apps for two novel medications used in the treatment of seizures and epilepsy, Oxtellar XR (oxcarbazapine extended-release tablets) and Trokendi XR (topiramate extended-release capsules).
Lilly and AstraZeneca announce alliance to co-develop potential treatment for Alzheimer’s...
Eli Lilly and AstraZeneca have announced an agreement to co-develop and commercialise AZD3293, an oral beta secretase cleaving enzyme (BACE) inhibitor currently in development as a potential treatment for Alzheimer's disease.
Covidien begins enrolment in two neurovascular clinical trials
Covidien has announced the start of enrolment in two clinical trials designed to further underscore the safety and effectiveness of the company's advanced neurovascular solutions.
IMRIS sterile wireless imaging coil and Monteris laser technologies combine for...
This combination brought improved visualisation and workflow in removing a patient's low-grade glioma brain tumour inside a VISIUS Surgical Theatre.
Codman Neuro gains exclusive rights to market and promote PulseRider in...
PulseRider is a minimally invasive device intended for use with embolic coils in the treatment of unruptured wide-neck intracranial aneurysms originating on or near a bifurcation.
Understanding the brain’s pulse shows promise for managing concussion
An article published in the Clinical Journal of Sport Medicine - "Detection of Concussion Using Cranial Accelerometry" - has shown that the Jan Medical Nautilus BrainPulse technology has detected a consistent pattern correlated with concussion.
Revive SE thrombectomy device approved in China, South Korea and Taiwan
The Revive SE Device from Codman Neuro is designed to ease navigation through small and tortuous blood vessels and arteries in the cerebral vasculature. The new device enables rapid restoration of blood flow to the brain during an acute ischaemic stroke.
Neuravi introduces collaborative clot research initiative at ESMINT
The Neuravi Thromboembolic Initiative brings together Neuravi engineers with clinicians and researchers in an effort to deepen the understanding of the mechanical properties of clot and occlusion dynamics, with the goal of improving the physician's ability to restore flow in acute ischaemic stroke.
Diplomat to distribute Plegridy (peginterferon beta-1a) for treatment of multiple sclerosis
Manufactured by Biogen Idec, Plegridy was approved on 15 August 2014 by the US Food and Drug Administration for the treatment of relapsing forms of multiple sclerosis (RMS).
GeNeuro phase 2a study reinforces novel approach to treat multiple sclerosis
GeNeuro has announced positive results from a one-year, open-label extension of a phase IIa study.
High accuracy for identification of traumatic intracranial haematomas with BrainScope, study...
The publication of an independent, prospective validation study has demonstrated the potential clinical utility of the BrainScope technology for the identification of acute traumatic intracranial haematomas in patients who present to hospital emergency departments.
National charity unveils big new ambitions for children with brain injury
The UK's leading charity for children with brain injury has launched a new three-year strategy, outlining ambitious plans to reach thousands more children and families across the UK.
NICE approves Tecfidera (dimethyl fumarate)
NICE has approved Tecfidera as an option for treating adults with active relapsing remitting multiple sclerosis.
Aggressive resection should be considered for paediatric epilepsy
Physicians from the University of California, San Francisco, USA, report that resection is safe and effective in children with pharmacoresistant focal epilepsy.
Alzheimer’s Prevention Registry reaches recruitment milestone of 40,000 individuals
The online Registry (www.endALZnow.org) aims to accelerate research by connecting healthy individuals who are committed to preventing Alzheimer's with scientists carrying out the studies.
Medtronic acquires Sapiens Steering Brain Stimulation
Medtronic has announced that it has acquired Sapiens Steering Brain Stimulation (Sapiens SBS) for approximately US$200 million in an all-cash transaction.
US Army funds phase II trial for treatment of PTSD with...
NeuroSigma has announced that the US Department of the Army, through the US Army Medical Research and Material Command, has funded a phase II clinical trial aimed at examining the use of external eTNS as treatment for post-traumatic stress disorder.
Highest level evidence indicates better outcomes when using iMRI for brain...
In a recently published article in the journal Neurosurgery the use of VISIUS intraoperative MRI (iMRI) in brain tumour surgery has been proven to result in complete tumour removal in more patients with glioma tumours.
Covidien acquires Reverse Medical Corporation
Covidien has announced that it has acquired Reverse Medical Corporation, a privately held medical device company focused on expanding the management of vascular disease. Financial terms of the transaction were not disclosed.
New non-invasive technique controls the size of molecules penetrating the blood-brain...
The innovative ultrasound approach uses acoustic pressure to let molecules through-may help treatment for central nervous system diseases like Parkinson's and Alzheimer's.
Utah team invents way to image brains of mice using small...
A University of Utah team discovered a method for turning a small, US$40 needle into a 3D microscope capable of taking images up to 70 times smaller than the width of a human hair.
New dementia research reveals overwhelming demand for “the value of knowing”
Three out of four people would want to know what kind of neurological disorder they had even if there was no cure, according to new global research from GE Healthcare.
Stemedica International announces pre-clinical data of Alzheimer’s study
The first pre-clinical mouse study using ischaemia-tolerant mesenchymal stem cells (itMSCs) shows over 30% decrease in amyloid beta (Abeta) plaques.
electroCore appoints Piper Jaffray to deal with growing interest from pharma
electroCore has appointed Piper Jaffray to assist in discussions with pharma companies who are interested in partnering on the commercialisation of its non-invasive vagus nerve (nVNS) stimulation therapy.
Low education, smoking, high blood pressure may lead to increased stroke...
Adult smokers with limited education face a greater risk of stroke than those with a higher education, according to new research in the American Heart Association's journal Stroke.
Plegridy (peginterferon beta-1a) approved in the USA for treatment of multiple...
Biogen Idec has announced that the US Food and Drug Administration has approved Plegridy (peginterferon beta-1a), a new treatment for people with relapsing forms of multiple sclerosis.
Enrolment in NeuroSTAT phase II study continues
Enrolment to the ongoing clinical phase IIa study with NeuroVive's drug candidate NeuroSTAT for treating patients with severe traumatic brain injury is continuing, and another two patients have been enrolled.
IMRIS intraoperative imaging moving system and coils patents granted in Japan
The patents cover control of effective MR imaging using a movable system; imaging coils where clarity is maintained when used while in position during X-ray use; and other components related to using both MR and X-ray imaging in the same environment.
NeuroPace RNS system receives approval from CMS for new Technology Add-On...
NeuroPace has announced that the Centers for Medicare & Medicaid Services (CMS) has approved New Technology Add-on Payments (NTAP) for the RNS system, the world's only commercially available implantable closed-loop responsive neurostimulator system.
AcuteCare Telemedicine and Colleton Medical Center partner to improve patient access...
AcuteCare Telemedicine continues to expand its presence in the Southeastern region with the addition of new client hospitals.
GE Healthcare’s Signa PET/MR 510(k) pending at FDA
GE Healthcare has announced that the first integrated, simultaneous, time-of-flight (TOF) capable, whole body Signa PET/MR is 510(k) pending at the US Food and Drug Administration.
Accenture and Philips announce proof of concept app for ALS patients
Royal Philips and Accenture have announced that they have developed proof of concept software connecting a wearable display to Emotiv Insight Brainware that could ultimately give more independence to patients with amyotrophic lateral sclerosis (ALS) and other neurodegenerative diseases.
Final patient treated in Neuralstem phase II ALS stem cell trial
Neuralstem has announced that the final patient was treated in its phase II trial using NSI-566 spinal cord-derived neural stem cells in the treatment of amyotrophic lateral sclerosis (ALS or Lou Gehrig's disease).
NeuroDerm announces eligibility for European Union centralised procedure for ND0612H
NeuroDerm has announced that the European Medicines Agency (EMA) has deemed ND0612H, its product candidate offering continuous delivery of levodopa/carbidopa (LD/CD) treatment for advanced Parkinson's disease, eligible for a European Union marketing authorisation application procedure ("centralised procedure").
Los Angeles medical team performs California’s first auditory brainstem implant surgery...
A Los Angeles team of scientists and surgeons reported that sound registered in the brain of a deaf Canadian boy for the first time after doctors activated a hearing device that had been surgically implanted in his brainstem.
New technology may improve visualisation of the brain during stroke treatment
New technology in the form of a magnetically-assisted remote-controlled catheter (MARC) which could allow physicians to see and assess brain tissue more clearly while treating a stroke may hold promise.
SNIS Foundation bestows first grant award for cutting-edge neurointerventional research
The Society of Neurointerventional Surgery (SNIS) Foundation recognised its first Seed Grant awardee with US$25,000 to fund a translational research project.
IMRIS receives CE mark for integrating latest generation MR scanners within...
IMRIS has announced that it has obtained regulatory CE mark for integrating the next generation MRI core technology into the VISIUS surgical theatre allowing for sales and marketing in the European Union.
Medtronic completes acquisition of Visualase
Medtronic has announced that it has completed the acquisition of Visualase, a privately held company based in Houston, USA, that develops and markets an FDA-approved MRI-guided laser and image guided system for minimally invasive neurosurgeries, including surgical thermal ablation.
NeuroPace RNS system will play key role in DARPA’s RAM programme
NeuroPace has announced its partnership with the Defense Advanced Research Projects Agency (DARPA) Restoring Active Memory (RAM) teams at the University of Pennsylvania and the University of California, Los Angeles (UCLA), USA to develop new treatments for memory deficits using neurostimulation.
Plegridy (peginterferon beta-1a) approved in the EU for treatment of multiple...
Biogen Idec has announced that the European Commission has granted marketing authorisation for Plegridy (peginterferon beta-1a) as a treatment for adults with relapsing-remitting multiple sclerosis.
US$1 million Career Development Award grant for glioblastoma research with ThermoDox...
The grant will support preclinical studies evaluating ThermoDox, heat-activated liposomal encapsulation of doxorubicin, in combination with High Intensity Focused Ultrasound (HIFU), for the treatment of brain tumours.
Scientists find new clues to brain’s wiring
New research provides an intriguing glimpse into the processes that establish connections between nerve cells in the brain. These connections, or synapses, allow nerve cells to transmit and process information involved in thinking and moving the body.
Study links enzyme to Alzheimer’s disease
Unclogging the body's protein disposal system may improve memory in patients with Alzheimer's disease, according to a study from scientists at Kyungpook National University in Korea published in The Journal of Experimental Medicine.
JAMA study: Stroke risk and death rates fall over past two...
Fewer Americans are having strokes and those who do have a lower risk of dying from them finds a new study led by Johns Hopkins Bloomberg School of Public Health researchers.
Making a mental match: Pairing a mechanical device with stroke patients
Georgia Tech researchers have created a mechanical device intended to help stroke victims. Their functional MRI-compatible hemiparesis rehab device creates a long latency stretch reflex at the exact time as a brain signal.
Stroke inpatient rehabilitation facilities yield better neurological outcomes than skilled nursing...
A demographic analysis has revealed that an association exists between discharge disposition and National Institutes of Health Stroke Scores (NIHSS) at 90-day follow-up.
Banner Alzheimer’s Institute partners with Novartis in new study of Alzheimer’s...
The study is to determine whether two investigational anti-amyloid drugs can prevent or delay the emergence of symptoms of Alzheimer's in people at particularly high risk for developing the disease at older ages.
C2N expands partnership with Washington University School of Medicine
The objective of this collaboration is to commercialise a clinical blood test for detecting the earliest stages of Alzheimer's disease as well as mild cognitive impairment.
Worldwide Alzheimer’s and dementia epidemic grows
While the global epidemic of Alzheimer's disease continues to grow, new data on lower incidence in the "youngest old" from developed countries in Europe and the United States suggest the possibility of reducing risk and/or preventing the disease, according to the results of several research studies.
Post-concussion ‘return to play’ decision for footballers should be made solely...
An editorial published in The Lancet Neurology calls for sports authorities to take into consideration the long-term neurological problems that repeated concussions can cause.
New data shows combination therapy is more effective than monotherapy in...
Data results found that combination therapy was more effective in removing clumps of amyloid-beta protein in the brain - a component that is thought to lead to Alzheimer’s disease - than use of one therapy.
Cataract surgery improves not only vision but cognition and quality of...
Cataract surgery for people with Alzheimer’s disease and other dementias not only improves vision but can slow decline in cognition and improve quality of life for both people with the disease and their caregivers, according to clinical trial results reported at the Alzheimer’s Association International Conference 2014 (AAIC) in Copenhagen, Denmark.
Potential Alzheimer’s disease risk factors and risk reduction strategies become clearer
Participation in activities that promote mental activity, and moderate physical activity in middle age, may help protect against the development of Alzheimer's disease and dementia in later life according to new research.
Looking back on 40 years of the Glasgow Coma Scale
A group of leading brain injury specialists look back on 40 years of the Glasgow Coma Scale and outline the continuing role of the scale in research and clinical practice, in a new Personal View published in The Lancet Neurology.
High stress, hostility, depression linked with increased stroke risk
Higher levels of stress, hostility and depressive symptoms are associated with significantly increased risk of stroke in middle-age and older adults, according to new research in Stroke.
New US patent for Barrel vascular reconstruction device
This new patent grants six claims surrounding the use of the company's Barrel vascular reconstruction device technology platform.
NIH funds phase II trial for treatment of ADHD with Monarch...
NeuroSigma has announced that the US National Institutes of Health (NIH) has awarded UCLA a grant that funds a phase II 90-subject paediatric clinical trial focused on the treatment of Attention Deficit Hyperactivity Disorder (ADHD) with the Monarch eTNS system.
Northwestern Medicine enrols first participant in study of device to treat...
According to doctors, the device could offer hope for patients with large, wide-neck brain aneurysms previously considered untreatable.
Cinnamon may be used to halt the progression of Parkinson’s disease
Neurological scientists at Rush University Medical Center have found that using cinnamon can reverse the biomechanical, cellular and anatomical changes that occur in the brains of mice with Parkinson's disease.
Less-invasive technique repairs life-threatening condition
A patient who underwent a less-invasive technique to repair an arteriovenous fistula was able to return to work just two weeks after surgery.
Brainlab announces launch of Right.Brain Foundation
Brainlab has announced the launch of the Right.Brain Foundation, which will provide medical technology and education to selected hospitals and public institutions in Southeast Asia, Africa, Central and South America.
Advances in trigeminal nerve stimulation featured at European congress on epileptology
NeuroSigma has announced top-line summaries of presentations made at the 11th European Congress on Epileptology in Stockholm, Sweden, related to the use of external trigeminal nerve stimulation (eTNS) in epilepsy.
UK researchers take new steps towards Alzheimer’s blood test
Researchers at King's College London and co-funded by Alzheimer's Research UK have announced a panel of 10 proteins that could form a blood test to predict those most likely to develop Alzheimer's.
SMC says yes to Lemtrada 12mg IV for adults with active...
The Scottish Medicines Consortium (SMC) has published its advice that Lemtrada has been accepted for use within NHS Scotland for the treatment of adult patients with relapsing-remitting multiple sclerosis (RRMS).
Mazor Robotics receives first order for Renaissance system with brain module
Mazor Robotics has announced that it has received the first order for its Renaissance system since the commercial launch of the brain surgery module.
New results show personalised brain tumour vaccine helps patients live longer
Patients newly diagnosed with glioblastoma multiforme (GBM) and treated with an experimental cancer vaccine made from the patient’s own tumour in addition to standard of care lived longer compared to those who received standard of care alone, according to new results from a study involving Northwestern Medicine researchers and released on 1 July.
New surgical tool provides hope for patients with inoperable deep bleeding...
The Apollo system is a new surgical tool that enables minimally invasive removal of deeply seated tissue and fluids in the brain during a single, efficient operation. With combined use of an endoscope and image guidance, the Apollo system allows decompression and removal of otherwise inoperable blood clots deep in the brain, among other uses.
Omega-3 fats may significantly reduce damage from stroke
New evidence shows that the omega-3 polyunsaturated fatty acids (PUFAs) found in seafood and marine oils, DHA and EPA, can significantly reduce damage from stroke in a mouse model when given immediately afterwards.
Scotland’s National Epilepsy Centre reports 100% success rate in first year
The William Quarrier Scottish Epilepsy Centre (WQSEC), a not-for-profit partnership between charity Quarriers and the NHS, welcomed 96 patients in the 12 months after opening in April 2013, its first annual report has revealed.
Agenus brain cancer vaccine shows extended survival in phase 2 final...
Agenus has announced final results from a phase 2 study showing that patients with newly diagnosed glioblastoma multiforme (GBM) who received Agenus' Prophage autologous cancer vaccine added to the standard of care treatment, lived nearly twice as long as expected.
electroCore’s funding US$10 million oversubscribed by all parties
The final tranche of US$15 million of the US$40 million was optional but following discussions between the investors this was not only made compulsory but increased by US$10 million to a total of US$50 million.
Successful combined approach to extensive cerebral venous sinus thrombosis with Penumbra...
A combined approach with the Penumbra aspiration system (Penumbra) and the Solitaire FR retrieval device (ev3, Covidien) has been successfully used in a patient with extensive superior sagittal sinus thrombosis and cortical venous thrombosis, according to a case report.
New brain-training app Brain+ launches in the UK
Brain+, a brain-training programme that outpaces existing apps in engagement, effectiveness and economy for users, is being launched in a brand new version on the 1 July.
Lemtrada approved in Argentina for treatment of multiple sclerosis
Genzyme has announced that Argentina's National Administration of Drugs, Food and Medical Technology (ANMAT) has approved Lemtrada (alemtuzumab) for adult patients with relapsing remitting multiple sclerosis (RRMS) with active disease defined by clinical or imaging features.
FDA clears Surgical Theater’s SNAP
The Surgical Navigation Advanced Platform (SNAP) integrates with operating room technology to provide advanced 3D capabilities and augmented reality, allowing surgeons to enhance their surgery performance and "see what cannot be seen".
China to build ‘brain database’
Chinese scientists are planning to build a "brain database" in a bid to identify clues to tackling cerebral diseases and related disorders.
AHA funds new research network aimed at preventing heart disease, stroke
The American Heart Association is funding a new research network to help people make behaviour changes to prevent heart disease and stroke, the two leading causes of death in the world.
Little or poor sleep may be associated with worse brain function...
Research published in PLOS ONE by researchers at the University of Warwick, UK indicates that sleep problems are associated with worse memory and executive function in older people.
New device allows brain to bypass spinal cord and move paralysed...
For the first time ever, a paralysed man has moved his fingers and hand with his own thoughts after an electronic neural bypass for spinal cord injuries that reconnects the brain directly to muscles, allowing voluntary and functional control of a paralysed limb. This innovation comes from a partnership between The Ohio State University Wexner Medical Center and Battelle.
Lemtrada (alemtuzumab) 12mg IV, shortlisted for the 2014 Prix Galien Innovative...
Genzyme has announced that it has been shortlisted for a Prix Galien award, in the Innovative Drug Award Category, for its multiple sclerosis therapy Lemtrada.
Pilot study shows gammaCore for the prevention of migraine is safe...
The study met its endpoint of safety, and also demonstrated a reduction in the number of headache days per month for patients using the active device.
New Neuro-Oncology chair for Ohio Clinical Trials Collaborative
The Ohio Clinical Trials Collaborative (OCTC) has appointed Jill Barnholtz-Sloan as chair of its Neuro-Oncology Working Group.
Neuralstem’s NSI-189 novel neurogenic compound shows significant effect in major depressive...
Neuralstem has announced that two sets of data from the NSI-189 clinical trial in major depressive disorder were reported at two recent academic conferences: American Society of Clinical Psychopharmacology Annual Meeting, and the International College of Neuropyschopharmacology Annual Meeting.
Imperial College London initiates study to investigate genetics of Parkinson’s disease
Researchers at the Imperial College London will study individuals with genetic mutations associated with Parkinson's disease as one of 32 clinical sites of the Parkinson's Progression Markers Initiative, a large-scale biomarker study sponsored by The Michael J Fox Foundation for Parkinson's Research.
Lingraphica introduces iPad app to help survivors of stroke and traumatic...
Lingraphica has introduced the iPad app component of TalkPath Therapy, its integrated cloud-based speech therapy solution for the millions of adults with aphasia write or comprehend language, and the clinicians who work with them to improve language skills.
Neuro Resource Group’s hospital presence continues to expand
Neuro Resource Group continues to expand the utilisation of its new InterX 900 ISSDE Pain Management System for post-surgical pain management in hospitals and surgery centres.
New Philips NeuroSuite reveals the brain’s vascular network like never before
Royal Philips has announced the introduction of NeuroSuite, a new integrated solution designed to support and enhance minimally invasive image-guided neurological interventions.
Gold for Airo Mobile Intraoperative CT in 2014 Medical Design Excellence...
Airo Mobile Intraoperative CT, developed and manufactured by Mobius Imaging and distributed by Brainlab, was awarded gold in the Radiological and Electromechanical Device category of the 17th Annual Medical Design Excellence Awards (MDEA) competition.
Researchers reveal combinatorial code for the developing brain
Researchers at the Allen Institute for Brain Science have mapped the development of the mouse brain from the embryo to the adult, creating a preliminary genetic key that allows them to pinpoint the age and location of regions of the developing brain.
Study proves navigated TMS fundamentally improves treatment and outcomes for brain...
A study published in the Oxford Journal Neuro-Oncology proved that integrating navigated transcranial magnetic stimulation (nTMS) into the surgical workflow to pre-operatively localise motor function has a significant impact on treatment and outcome.
Neurim Pharmaceuticals announces publication of positive effects of add-on Circadin in...
Neurim Pharmaceuticals has announced publication of the results from an exploratory phase 2 randomised placebo-controlled clinical trial evaluating the safety and efficacy of add-on Circadin (Prolonged Release melatonin 2mg) to standard therapy in Alzheimer’s disease patients.
Foreign body retrieval could be new indication for Trevo ProVue in...
A case report has described the novel use of a stent for the removal of a foreign body from the cerebrovascular system.
New insight into how the brain regulates its blood flow
In a new study published online in the Journal of the American Heart Association, researchers at Columbia Engineering report that they have identified a new component of the biological mechanism that controls blood flow in the brain.
Floridian Community Bank supports NSU Concussion Management Clinic
Floridian Community Bank has announced that it will sponsor the Nova Southeastern University Concussion Management Clinic, one of the largest community-based sport concussion initiatives in the state of Florida.
Northwestern Medicine brings hope to patients with severe epilepsy
Specialists at Northwestern Medicine’s Comprehensive Epilepsy Center recently started using a new surgical procedure that could change the lives of epileptics who are unable to control their seizures with medication.
Vantage one-year data shows highly significant improvement in motor scores for...
Patients with Parkinson's disease treated with the Vercise deep brain stimulation system (Boston Scientific) demonstrated a highly significant and consistent improvement in motor scores, according to the latest one-year data.
Alzheon to participate in Global Dementia Legacy Meeting
Expanding on the initiatives of the G8 Dementia Summit held in December 2013, the Global Dementia Legacy Meeting will propose ways to increase and apply investments in dementia.
BrainCare promotes brain wellness after stroke and traumatic brain injury
NeuroTrax has announced the global availability of BrainCare, a cognitive assessment and report solution to aid clinicians in advancing brain wellness of stroke survivors and traumatic brain injury patients.
Parkinson’s disease trial shows that for long-term treatment levodopa is better...
For long-term treatment of newly diagnosed Parkinson's disease, the old drug levodopa provides better mobility and a higher quality of life than the two main alternatives, dopamine agonists and monoamine oxidase type B inhibitors, according to a study published in The Lancet.
Study results demonstrate advantages of Vercise
The CUSTOM-DBS clinical study demonstrated that shorter stimulation pulses may offer a clinical advantage over deep brain stimulation therapy using conventional pulses.
Brain implant offers hope for epilepsy patients
Physicians at Stanford Hospital & Clinics now offer an implantable therapeutic device, designed to detect and treat seizures, for certain patients with epilepsy.
FDA approves Apollo Onyx microcatheter
The new microcatheter is designed to mitigate the technical challenges of catheter retrieval during Onyx liquid embolic system embolisations of brain arteriovenous malformations.
Trial shows gammaCore is effective in reducing cluster headaches
In a multicentre, randomised, trial across Europe, electroCore's non-invasive vagus nerve stimulation therapy was found to meet its primary endpoint of statistical significance in reducing the number of cluster headache attacks when compared with the standard of care.
FDA approves expanded label for Azilect
The US Food and Drug Administration (FDA) has expanded the indication for Azilect (rasagiline tablets) from monotherapy and adjunct to levodopa to now include adjunct to dopamine agonists. The new indication reflects that Azilect can be used alone or in combination with other Parkinson's disease medications.
FDA IDE approval for PulseRider
The IDE allows Pulsar Vascular to begin a multicentre clinical trial in support of a humanitarian device exemption (HDE) to evaluate the PulseRider for US approval for wide neck aneurysms at or near a bifurcation of the basilar tip or carotid terminus.
MRI-guided laser procedure provides alternative to epilepsy surgery
For patients with mesial temporal lobe epilepsy (MTLE) that can't be controlled by medications, a minimally invasive laser procedure performed under MRI guidance provides a safe and effective alternative to surgery, suggests a study in the June issue of Neurosurgery.
Enterprise stent was found to be a safe and effective revascularisation...
Researchers report that in a prospective study, the Enterprise stent (Codman) was found to be a safe and effective when used for primary stenting. The findings were published in the Journal of NeuroInterventional Surgery.
USA’s first mobile stroke unit successfully transports first patient
The University of Texas Health Science Center at Houston Medical School, in partnership with Memorial Hermann-Texas Medical Center, USA, has announced that the UTHealth Mobile Stroke Unit has successfully transported and helped save the life of its very first patient.
A new approach to Alzheimer’s disease research
The Allen Institute for Brain Science, Seattle, USA, is embarking on the first effort to map connectivity patterns across the whole brain in mouse models of Alzheimer’s disease, through its recent award of a US$3.4 million grant over five years from the National Institute on Aging of the National Institutes of Health.
Breakthrough in Alzheimer’s disease: AFFiRiS halted clinical progression in Alzheimer patients...
The results of the phase II trial show an impressive therapeutic effect of AD04 and make it the first ever compound demonstrating clinical and biomarker evidence consistent with disease modification of Alzheimer's disease.
Mayo Clinic moves small-molecule drugs through blood-brain barrier
Researchers at Mayo Clinic have demonstrated in a mouse model that their recently developed synthetic peptide carrier is a potential delivery vehicle for brain cancer chemotherapy drugs and other neurological medications. The findings appear in PLOS ONE.
Clinical trial tests possible benefits of brain stimulation on hand and...
Researchers at Shepherd Center, Atlanta, USA, are studying whether stimulating the brain before rehabilitation could yield greater gains in motor function for people recovering from stroke. Researchers believe that the new approach could change common practices of care for certain stroke patients.
Constant Therapy launches iPad solution for people with traumatic brain injury,...
Designed to be used independently by anyone seeking to improve brain function or in conjunction with a clinician as an integral part of therapy, this new mobile solution integrates ground-breaking research on brain plasticity and rehabilitation from Boston University (USA) and other leading institutions.
FDA clears Night Shift therapy
Advanced Brain Monitoring has announced that the US Food and Drug Administration (FDA) has granted 510(k) clearance for Night Shift, a therapy for positional obstructive sleep apnoea.
NeuroBlate system for minimally invasive ablation of brain lesions to be...
At the biennial meeting of the American Society of Stereotactic and Functional Neurosurgery (31 May-3 June; Washington, DC, USA) Gene Barnett, director of the Rose Ella Burkhardt Brain Tumor and NeuroOncology Center at the Cleveland Clinic, will present his experience utilising NeuroBlate system (Monteris Medical) laser ablation in patients with brain tumours and radiation necrosis.
Angiochem to present positive central nervous system and peripheral anti-tumour activity...
Phase 1 and phase 2 clinical studies demonstrated promising signs of both central nervous system and peripheral anti-tumour activity of ANG1005 in patients with brain metastases including breast cancer patients.
Lemtrada resubmission accepted for review by FDA
Genzyme has announced that the US Food and Drug Administration (FDA) has accepted for review the company's resubmission of its supplemental Biologics License Application (sBLA) seeking approval of Lemtrada (alemtuzumab) for the treatment of relapsing forms of multiple sclerosis. A six-month review period has been assigned for the Lemtrada sBLA.
Symbis Surgical System named to 2014 Space Technology Hall of Fame
IMRIS has announced that the SYMBIS Surgical System - the second generation of the neuroArm / Symbis programme - is a 2014 inductee into the Space Technology Hall of Fame. The Space Foundation honour is directed at technologies originally developed for space exploration that are being transformed into products to help improve the quality of life on Earth.
Teva and Active Biotech to continue with the development of Nerventra...
Teva Pharmaceutical Industries and Active Biotech have announced that the Committee for Medicinal Products for Human Use (CHMP) confirmed its January 23, 2014 opinion to recommend against approval for the treatment of relapsing-remitting multiple sclerosis in the European Union at this time.
NICE recommends Lemtrada (alemtuzumab) 12mg IV for the treatment of active...
The National Institute for Health and Care Excellence (NICE) has issued final guidance recommending that Lemtrada should be reimbursed on the National Health Service (NHS), as an option for treating adults with active relapsing remitting multiple sclerosis.
Older migraine sufferers may have more silent brain injury
Previous studies have indicated migraine could be an important stroke risk factor for younger people. A new study published in Stroke now says that older migraine sufferers may be more likely to have silent brain injury.
Inovio Pharmaceuticals acquires early stage DNA therapies to treat Alzheimer’s and...
Inovio Pharmaceuticals has announced that it has acquired worldwide rights (excluding China) for early preclinical therapies addressing Alzheimer's disease and multiple sclerosis based on the academic research of Bin Wang, a professor at Fudan University's Shanghai Medical College.
GE Healthcare and CorTechs Labs announce strategic agreement to co-market NeuroQuant
NeuroQuant enables the acquisition of volumetric brain image data and automatically conducts accurate and consistent measurements of cortical and subcortical volumes targeted at identifying evidence of neurodegeneration. This innovative solution will provide neurologists and radiologists with objective support for their clinical impression.
FDA clears SpringTMS migraine treatment device
SpringTMS is the first medical device available to patients in the USA for the acute treatment of pain associated with migraine headache with aura. The device is currently CE marked in Europe and is available to patients in the UK.
Significant milestones set in world’s most detailed brain imaging study to...
As part of the global effort to defeat dementia, around 100,000 participants are expected to be part of the world's most comprehensive imaging project over the next five years, following the launch of a feasibility study.
Neurotrope collaborates with Paul Wender and Stanford University on bryostatin analogues
Neurotrope has signed an agreement with Stanford University to study and, along with Paul Wender, investigate certain analogs of bryostatin, referred to as "bryologs", as potential clinical candidates for the treatment of various neurological disorders.
Barnes-Jewish Hospital neurosurgeons treat 1,000th patient using VISIUS iMRI
IMRIS has announced that Barnes-Jewish Hospital in St Louis is the first US hospital to use VISIUS iMRI for more than 1,000 procedures. This clinical experience and published evidence by the neurosurgical team has shown that intraoperative MRI is an effective tool for improving results and outcomes for patients undergoing brain surgery.
Keystone heart raises US$14m in series B funding for cerebral protection...
Keystone Heart has raised US$14 million in series B funding for cerebral protection devices. By protecting all brain territories, the TriGuard cerebral protection device is designed to minimise the risk of cerebral damage during transcatheter aortic valve implantation (TAVI) and other cardiovascular procedures.
Increase in hospitalisation for subarachnoid haemorrhage also sees decline in mortality...
Tobias Pilgaard Ottosen (Department of Clinical Epidemiology, Aarhus University Hospital, Denmark), in a presentation at European Stroke Conference (Nice, France, 6-9 May) told delegates that, over a 30-year period, first-time hospitalisation for subarachnoid haemorrhage has increased.
Cooling system may have neuroprotective effects in acute stroke
A poster presented at the European Stroke Conference (6-9 May 2014, Nice, France) by Robbert-Jan van Hooff (Department of Neurology, Universitair Ziekenhuis Brussel, Brussels, Belgium) and colleagues reports that surface cooling of the neck with a surface cooling system could contribute to therapeutic hypothermia and neuroprotective effects in acute stroke.
Long-term comparative effectiveness study of VNS in patients with refractory epilepsy...
Cyberonics has announced positive results from the Pulse (Open prospective randomised long-term effectiveness) study of vagus nerve stimulation (VNS) therapy in patients with refractory epilepsy conducted at 28 sites in Europe and Canada and published in the journal Epilepsia.
Sleep researchers at SRI International identify promising new treatment for narcolepsy
Neuroscientists at SRI International have found that a form of baclofen, a drug used to treat muscle spasticity, works better at treating narcolepsy than the best drug currently available when tested in mice.
Neurosurgery chair appointed at Henry Ford Hospital
Neurosurgeon Steven N Kalkanis has been appointed chair of the Department of Neurosurgery and co-director of the Neuroscience Institute at Henry Ford Hospital in Detroit, USA.
New MRI CryoProbe delivers highest sensitivity in rat brain in vivo...
At the joint ISMRM-ESMRMB 2014 meeting (10-16 May, Milan, Italy), Bruker announced a new phased array MRI CryoProbe for in vivo neuroimaging that delivers unparalleled access to rat brain microstructure and biochemistry, previously unresolvable in in vivo imaging of the rat.
Non-drug TMS therapy shows favourable outcomes compared to oral anti-depressants
Neuronetics announced a new analysis of data at the annual meeting of the American Psychiatric Association that shows transcranial magnetic stimulation (TMS) administered with the NeuroStar TMS therapy system resulted in greater symptom improvement than next-choice conventional anti-depressant medication.
First treatment for babies with ultra-rare spectrum of autoinflammatory diseases now...
Anakinra (Kineret), an interleukin-1 (IL-1) inhibitor, is now available in the UK for treatment of CAPS in children and adults. Anakinra is the first CAPS treatment licensed for use in infants as young as eight months by the Medicines and Healthcare Products Regulatory Agency (MHRA).
Neurosurgical design nominated for business innovation award
Known as Rowena, the neurological simulator was shortlisted from over 950 entries in the FSB WorldPay UK Business Awards which take place at the Waldorf Hotel in London on 21st May. Rowena was created by consultant neurosurgeon Richard Ashpole, founder of Neurodesign.
Novel laser device used for brain surgery
Monteris Medical announced that their novel magnetic resonance imaging (MRI)-guided laser technology, the NeuroBlate system, was recently applied by neurosurgeons at UC San Diego Health System to a malignant tumour deep inside a patient's brain.
Alzheimer’s Drug Discovery Foundation raises US$1.1M for new clinical trial in...
The US$1.1 million was generated during the "Fund a Scientist" auction at its Eighth Annual Connoisseur’s Dinner on May 1, 2014, and awarded to Jeffrey Cummings, director of the Cleveland Clinic Lou Ruvo Center for Brain Health in Las Vegas, Nevada and Cleveland, Ohio, USA. Cummings will investigate rasagiline, an FDA-approved treatment for Parkinson’s disease with the potential to be the first drug to slow the course of Alzheimer’s disease.
Genzyme announces multi-year multiple sclerosis research collaboration
The research collaboration with Cleveland Clinic will be focused on developing new therapeutic approaches to the treatment of multiple sclerosis.
Two studies on the treatment of migraine headache with non-invasive vagus...
At the American Association of Neurology (AAN) meeting in Philadelphia, USA, two separate studies showed that electroCore's non-invasive vagus nerve stimulation (nVNS) therapy was effective in helping patients with different types of migraine.
Lpath lands fast-track SBIR grant from the NIH for traumatic brain...
Lpath has received official notification from the National Institutes of Neurological Diseases and Stroke (NINDS) that it has been awarded a US$1.7 million, 2.5-year grant to develop its antibody therapeutic, Lpathomab, which is directed against the bioactive lipid lysophosphatidic acid (LPA) for the treatment of traumatic brain injury.
Advance study completes enrolment for deep brain stimulation for Alzheimer’s disease
Functional Neuromodulation has completed enrolment for the Advance study. Forty-two North American patients with mild Alzheimer's have had a deep brain stimulation system implanted to evaluate the safety and potential clinical benefit of deep brain stimulation of the fornix.
Phase II study of Trimesta for relapsing-remitting multiple sclerosis yields positive...
Synthetic Biologics reports that the University of California, Los Angeles (UCLA) School of Medicine (USA) has announced preliminary, positive top-line data from the phase II clinical trial evaluating Trimesta (oral estriol), an oral, once-daily treatment for relapsing-remitting multiple sclerosis in women.
Mechanism of action of electroCore’s non-invasive vagus nerve stimulation therapy published...
In a paper published in the Journal of Pain it was reported that electroCore's non-invasive vagus nerve stimulation (nVNS) therapy can significantly reduce excessive glutamate levels in key regions of the central nervous system of animals sensitised to experience migraine-like symptoms.
Two-year study finds that vitamin E from palm oil helps to...
The results of a two-year human clinical study have been published in the American Heart Association journal, Stroke. The results show that vitamin E tocotrienols derived from Malaysian palm oil may support white matter health by weakening the progression of white matter lesions.
Study examines risk of early death for people with mild cognitive...
One of the first studies to look at a relationship between death and the two types of mild cognitive impairment suggests that people who have thinking problems but their memory is still intact might have a higher death rate in a period of six years compared to those who have no thinking or memory problems.
Lpath and Walter Reed Army Institute for Research begin efficacy study...
Lpath and scientists from the Center for Military Psychiatry and Neuroscience, Walter Reed Army Institute of Research (WRAIR), USA, have initiated a study on the use of Lpathomab, an antibody to lysophosphatidic acid (LPA), in the treatment of brain injury induced by blast overpressure.
AdvaStim enters neuromodulation device market
AdvaStim has announced that it has entered the research and development market with a new hardware/software solution for neuromodulation therapy developers.
Tampa General Hospital launches advanced intraoperative MRI use with first cases
IMRIS has announced that a University of South Florida neurosurgical team at Tampa General Hospital (TGH), USA, completed initial cases last week using its recently installed VISIUS intraoperative MRI. The first case was a right parietal high-grade glioma.
Legal safeguards needed for brain-boosting devices
Researchers from the Oxford Martin School, University of Oxford, UK, are calling for the regulation of a new breed of devices designed to enhance the brain's performance. According to Oxford Martin School, these devices, because of European legislation, only receive basic product safety requirements.
Therapies based on newly-discovered GAIM could combat neurodegenerative disorders
Researchers from NeuroPhage Pharmaceuticals have engineered a series of molecules with the potential to treat most neurodegenerative diseases that are characterised by misfolded proteins, such as Alzheimer's, Parkinson's and Huntington's diseases, according to a press release.
Carotid stenosis without warning may signal memory and thinking decline
For the first time, researchers have demonstrated that asymptomatic carotid stenosis may be linked to problems in learning, memory, thinking and decision-making, compared to people with similar risk factors but no carotid stenosis.
Study reinforces value of high-field iMRI in brain tumour resection
IMRIS has announced that a study published in the journal Neurosurgery by the neurosurgical team at Cleveland Clinic (USA) adds to growing clinical evidence which validates use of high-field intraoperative MRI (iMRI) as an effective tool for maximising the amount of surgical resection of gliomas.
Functional brain imaging reliably predicts which vegetative patients have potential to...
A functional brain imaging technique, positron emission tomography (PET), is a promising tool for determining which severely brain damaged individuals in vegetative states have the potential to recover consciousness, according to new research published in The Lancet.
Scottish Medicines Consortium approves Tecfidera for multiple sclerosis
The Scottish Medicines Consortium (SMC) has approved Tecfidera (dimethyl fumarate) as a first-line oral treatment for people with relapsing-remitting multiple sclerosis.
World’s first high-field MRI-guided radiation therapy system being installed at Dutch...
Elekta, Royal Philips and University Medical Center Utrecht (the Netherlands) announced that the initial components of the world's first high-field magnetic resonance imaging (MRI) guided radiation therapy system are being installed at University Medical Center Utrecht.
First patient enrolled in registry assessing Vercise deep brain stimulation system
Boston Scientific has enrolled the first patient in a new registry to evaluate clinical outcomes and the economic value of the Vercise deep brain stimulation system in patients with Parkinson's disease.
Covidien implements voluntary recall of its Pipeline embolisation device and Alligator...
Covidien has announced that it has notified customers of a voluntary recall to address an issue with certain lots of its Pipeline embolisation device and Alligator retrieval device where the polytetrafluoroethylene (PTFE) coating applied to the delivery wire could delaminate and detach from the devices.
CE mark approval for Airo mobile intraoperative CT
Brainlab and Mobius Imaging announced CE mark approval for Airo mobile intraoperative computed tomography. The CE mark allows Brainlab to begin installation of the systems sold in the European Union.
Monarch eTNS system receives market approval from Australian Therapeutic Goods Administration
NeuroSigma announced that it has received approval from the Therapeutic Goods Administration (TGA) to market its Monarch eTNS (external trigeminal nerve stimulation) system in Australia. The approval allows NeuroSigma to market the Monarch eTNS system for the adjunctive treatment of drug-resistant epilepsy in patients nine years of age and older.
Cognition Therapeutics receives a US patent for Alzheimer’s drug
Cognition Therapeutics has received a US patent for a drug to fight Alzheimer's disease. The patent describes the discovery of several candidate drug molecules which can stop the effects of the brain protein that appears to play a major role in development of Alzheimer’s disease.
NeuroVascular Quality Initiative launches first procedure module: Acute Ischaemic Stroke
The Society of NeuroInterventional Surgery and M2S announced the launch of the NeuroVascular Quality Initiative, a multi-module registry system designed to track performance and outcomes data across a variety of neurovascular conditions. The launch debuts the Acute Ischaemic Stroke module, which will be used to track and benchmark clinical performance at the centre level and nationally.
IMRIS iMRI and ClearPoint neuro intervention system utilised for first paediatric...
The combination of the IMRIS Visius surgical theatre and the ClearPoint neuro intervention system provided continuous real-time visualisation and guidance throughout a neurosurgical intervention to treat a brain tumour.
Antioxidant shows promise in supporting treatment for ALS
Researchers at The Center of Free Radical and Biomedical Research at Facultad de Medicina, Universidad de la Republicain Uruguay announced the discovery that the antioxidant MitoQ prolonged the lifespan and health-span of mice suffering from amyotrophic lateral sclerosis-like (ALS) disease, and successfully improved functional parameters related to muscular strength and reversed mitochondrial damage in nervous and muscle tissue.
FDA approval for Neuraceq (florbetaben F18 injection)
Neuraceq is indicated for Positron Emission Tomography (PET) imaging of the brain to estimate beta-amyloid neuritic plaque density in adult patients with cognitive impairment who are being evaluated for Alzheimer's disease and other causes of cognitive decline.
Morphology and interval growth are predictive factors of intracranial aneurysm rupture
In a study conducted by William Mehan et al, it was found that for patients with unruptured intracranial aneurysms, multilobulated morphology and interval growth of the unruptured aneurysms are "characteristics predictive of a higher risk of subsequent rupture during conservative CT angiography (CTA) follow-up."
ADAPT a safe and simple technique for ischaemic stroke thrombectomy
A direct aspiration first pass technique (ADAPT) with a large bore aspiration catheter has been shown to be "fast, safe, simple and effective method" for acute ischaemic stroke thrombectomy. The results were published in the Journal of of NeuroInterventional Surgery.
Statins slow the progression of advanced multiple sclerosis
In a two-year clinical trial involving 140 patients with secondary progressive multiple sclerosis, the drug simvastatin slowed brain shrinkage, which is thought to contribute to patients' impairments.
Prodigy spinal cord stimulation system gets CE mark approval
St Jude Medical has received CE mark approval for its Prodigy chronic pain system with burst technology, the first and only implantable neuromodulation system that delivers burst stimulation.
Functional brain imaging can provide better diagnosis and treatment monitoring in...
A new paper published by a joint US and Canadian team in PLOS One suggests SPECT (single photon emission computed tomography)-a functional brain imaging modality that produces images of blood flow to the brain, showing areas of over or under-activity-is useful in both identifying and guiding treatment for those with traumatic brain injuries.
Data from a study of PBA symptoms in veterans with mild...
Avanir Pharmaceuticals announced the presentation of results from a first-of-its-kind study of pseudobulbar affect (PBA) symptoms in veterans with mild traumatic brain injury conducted in collaboration with the Department of Veterans Affairs and Evidera.
FDA allows marketing of first medical device to prevent migraine
The US Food and Drug Administration (FDA) has approved marketing of Cefaly, the first device as a preventative treatment for migraine. This is also the first transcutaneous electrical nerve stimulation (TENS) device specifically authorised for use prior to the onset of pain.
Dartmouth-Hitchcock Medical Centre inaugurates Visius surgical theatre
IMRIS announced that neurosurgeons at Dartmouth-Hitchcock Medical Centre in Lebanon, USA, have completed several cases to inaugurate use of intraoperative MRI (iMRI) within the Visius surgical theatre inside the hospital's Centre for Surgical Innovation.
Final results of Neuralstem phase I stem cell trial in ALS...
The final results from the phase I safety trial using NSI-566 spinal cord stem cells in the treatment of amyotrophic lateral sclerosis (ALS) were published in the peer-reviewed journal, Annals of Neurology.
Stroke survivors may lose month of healthy life for 15-minute delay...
Every 15-minute delay in delivering a clot-busting drug after stroke robs survivors of about a month of disability-free life, according to a new study in the American Heart Association journal Stroke.
Blood biomarkers could help guide return to play for athletes after...
In a recent study it was found that ice hockey players in Sweden with a sports-related concussion had higher levels of the blood biomarker total tau (T-tau), which suggests the central nervous system protein may be a tool for diagnosing concussions and making decisions about when players can return to play.
Philips celebrates World Sleep Day to raise awareness of the benefits...
Philips has launched a campaign for 2014 which highlights sleep as a powerful engine for all our daily activities.
Study finds high utilisation of neuroimaging for headaches despite guidelines
A recent study has found that neuroimaging for headaches is frequently ordered by physicians during outpatient visits, despite guidelines that recommend against such routine procedures.
Significant head and neck injury risk associated with extreme sports
A new study shows that extreme sports such as snowboarding and skateboarding present high risk for head and neck injuries, including concussions. The findings support need for greater education and advocacy efforts for safer equipment.
Clinical study finds electroCore’s non-invasive vagus nerve stimulation effective in treating...
A study reported in the International Headache Society's official journal Cephalalgia found that the use of electroCore's non-invasive vagus nerve stimulation therapy may be an effective and well tolerated treatment for migraine in certain patients.
Study finds no link between football play and neurocognitive function in...
A new study presented at the 2014 Annual Meeting of the American Academy of Orthopaedic Surgeons (AAOS) found no link between neurocognitive function and years of football play in adolescent athletes.
24-month data show back and leg pain scores are reduced with...
The 24-month results of a prospective European clinical study using Nevro Corp's Senza system delivering HF10 therapy were published in the journal Pain Medicine. The study was conducted in the UK and Belgium with 65 of the 72 implanted patients (90%) being available for data collection at two years.
Enrolment begins in a second phase II study of ND0612 for...
NeuroDerm announced that enrolment of patients is ongoing in its second phase II clinical trial of ND0612, a novel drug formulation for the treatment of Parkinson's disease.
SMC says yes to Aubagio (teriflunomide)
The new once daily, first-line oral treatment can be offered as an alternative to currently available injectable treatment options for people with an active relapsing remitting form of multiple sclerosis.
Phase III GBM brain cancer trial gets recommendation to continue based...
Northwest Biotherapeutics has announced that the Data Safety Monitoring Board has made an unblinded review of the safety data for the company's ongoing international Phase III Glioblastoma multiforme (GBM) Trial, and has recommended that the trial continue as planned.
Flow diverters safe and feasible in ruptured blood blister-like aneurysms
Published in the Journal of NeuroInterventional Surgery, the results of a small study of 11 patients has demonstrated that Silk flow diverter stents (Balt Extrusion) could be a safe and feasible treatment for fragile, difficult-to-treat blood blister-like aneurysms.
Aethlon Medical and Exosome Sciences announce brain research discoveries
The brain research discoveries could have implications in the diagnosis, monitoring and treatment of Alzheimer's disease, chronic traumatic encephalopathy and traumatic brain injury.
Stroke patients treated at stem cell research centre
Stem cell research institute of STC Life and 97.7 B&H Clinic successfully completed mesenchymal stem cell treatment for stroke patients.
D-Pharm reports successful interim analysis of phase II clinical study
D-Pharm has reported the successful interim analysis of its phase II clinical study of THR-18 in acute stroke patients treated with the thrombolytic drug tPA.
US clinical site opens for expansion of CEL-SCI’s global phase III...
CEL-SCI Corporation announced its phase III head and neck cancer clinical trial of its investigational cancer immunotherapy treatment Multikine (Leukocyte Interleukin, injection) has activated its first US clinical trial expansion site at 21st Century Oncology in Greenville, North Carolina, USA.
Forest Laboratories submits new drug application for memantine extended release and...
Forest Laboratories and Adamas Pharmaceuticals announced Forest's submission of a new drug application to the US Food and Drug Administration for a fixed-dose combination of memantine HCl extended release and donepezil HCl for the treatment of moderate to severe dementia of the Alzheimer's type.
New published paper shows efficacy of Lpathomab in traumatic brain injury...
Lpath has brought scientists one step closer to finding a potential treatment for traumatic brain injury with a recent publication showing that Lpathomab, an anti-lysophosphatidic acid antibody, reverses much of the damage caused by trauma to the nervous system.
Phase I/II clinical trial for ITI-007 in healthy geriatric subjects and...
Intra-Cellular Therapies announced the initiation of ITI-007-200, a Phase I/II clinical trial designed to evaluate the safety, tolerability and pharmacokinetics of low doses of its lead drug candidate, ITI-007, in healthy geriatric subjects and in patients with dementia, including Alzheimer’s disease.
Public to get a glimpse into cutting edge brain research
University of Leicester scientists will showcase their research findings as part of Brain Awareness Day on Wednesday, 12 March
Study shows that teenagers treated for headaches were prescribed opioids almost...
Clinicians prescribed opioids for almost half of the teenagers they treated for headache when medications, such as aspirin, ibuprofen and naproxen, are recommended as first-line therapies, according to a study in the Journal of Adolescent Health.
New partnerships designed to advance Parkinson’s and dystonia care
The aim of these partnerships between Dystonia Europe and the European Parkinson's Disease Association and Boston Scientific is to increase awareness, understanding of solutions and to take initiatives in the worldwide fight against Parkinson's disease and dystonia, two areas of high unmet need.
Piramal Imaging announces EU approval of NeuraCeq
NeuraCeq (florbetaben 18F) has been approved for PET imaging of beta-amyloid neuritic plaque density in the brain.
Study shows that MS antibody may be detectable years before symptoms...
An antibody found in the blood of people with multiple sclerosis may be present long before the onset of the disease and its symptoms, according to a recent study that will be presented at the American Academy of Neurology's 66th Annual Meeting in Philadelphia, USA, April 26 to May 3, 2014.
Takeda joins the Global CEO Initiative on Alzheimer’s disease
The Global CEO Initiative (CEOi) on Alzheimer's Disease announced that Takeda Pharmaceutical Company will join the CEOi as part of its effort to spur innovations in research and accelerate a means of prevention for Alzheimer's and dementia, building momentum behind the G8's commitment to stop the disease by 2025.
Vitae earns US$14 million milestone payment from Boehringer Ingelheim in Alzheimer’s...
Vitae Pharmaceuticals announced that it has earned a US$14 million milestone payment from Boehringer Ingelheim for that company’s initiation of a Phase I clinical trial in the Vitae’s beta-secretase (BACE) inhibitor programme for Alzheimer’s disease.
Stryker launches Trevo XP ProVue retrieval system
Stryker has launched the Trevo XP ProVue retrieval system, a second generation fully visible stent retriever to treat acute ischaemic stroke.
Michael J Fox Foundation to fund the use of GlycanMap technology...
Ezose Sciences has announced that it has received a grant from the Michael J Fox Foundation for Parkinson's Research to apply Ezose's GlycanMap technology to investigating the role of the sugars known as glycans in Parkinson's disease.
STC Life successfully treats patients with Parkinson’s disease at stem cell...
The stem cell research institute of STC Life and 97.7 B&H Clinic successfully completed mesenchymal stem cell therapy for patients with Parkinson's disease.
Reverse Medical announces CE mark of UNO neurovascular embolisation system
Reverse Medical Corporation has announced the initial clinical use of their UNO neurovascular embolisation system for intracranial use. The device has been granted European Union CE mark approval to obstruct blood flow in the neurovasculature.
Clinical study focused on innovations in Parkinson’s disease biomarker discovery announced
Berg and the Parkinson's Institute and Clinical Centre has announced the next phase in their ongoing partnership, focused on identifying potential biomarkers that may lead to breakthroughs in the research, diagnosis, and treatment of Parkinson's disease.
IMRIS launches world’s first MR-safe and CT-compatible neurosurgical horseshoe headrest
The IMRIS horseshoe headrest provides non-rigid head positioning for paediatric and adult patients during brain surgery and intraoperative imaging in the VISIUS surgical theatre.
New study results demonstrate value of Cerox cerebral oxidation monitor
Ornim Medical has announced that new study results demonstrate the feasibility of the Cerox cerebral oxidation monitor for measuring regional cerebral tissue oxygenation in patients with severe traumatic brain injury.
FDA clears next generation VISIUS surgical theatre
IMRIS has announced US Food and Drug Administration (FDA) clearance of the newest generation VISIUS surgical theatre which integrates Siemens' latest high-field magnetic resonance (MR) scanners for use during neurosurgical procedures.
New stroke therapy produced significant gains in motor function post-stroke
New stroke therapy, using a combination of non-invasive navigated transcranial stimulation along with occupational therapy, has been found to produce significant gains in motor function post-stroke, with an average of 50% of stroke survivors recovering full use of their arm.
MIT professor joins Constant Therapy’s Scientific Advisory Board
Constant Therapy, a developer of a cloud-based iPad solution enabling people with cognitive, language, communication and learning disorders to access science-based brain therapies, has announced that John Gabrieli, Massachusetts Institute of Technology (MIT) Brain and Cognitive Sciences professor, has joined the company's Scientific Advisory Board.
FDA classifies worldwide voluntary correction notice on Trufill at Class I...
Codman Neuro announced that the US Food and Drug Administration (FDA) has classified the recently initiated medical device correction notice related to the Trufill n-BCA liquid embolic system as a Class I recall.
Intracranial carotid artery atherosclerosis associated with increased stroke risk
A build-up of plaque in the carotid artery above the neck was associated with an increased risk of stroke for older white patients in a recent study.
InspireMD reports successful implantation of the new CGuard carotid embolic system...
InspireMD has announced that its new CGuard carotid embolic protection system, designed to trap the debris that can travel downstream after a patient is treated with traditional stenting methods, has been successfully implanted in recent procedures.
Brain Research Foundation announces 2014 Scientific Innovations Awards
The Brain Research Foundation has announced the 2014 Scientific Innovations Awards to fund ground-breaking work in Parkinson's disease, epilepsy, Hurler syndrome and autism.
MRI increases the incidence of extra-axial intracranial tumour diagnosis
A Norwegian study has found that cerebral, regional magnetic resonance imaging (MRI) can lead to an increase in incidence rates of intracranial tumour diagnosis.
Vilcek Foundation honours renowned neuroscientist
British-born neuroscientist honoured by Vilcek Foundation.
The ALS Association announces new grants to investigate the causes of...
Five new grants have been awarded to investigate the causes of and treatments for amyotrophic lateral sclerosis
Tecfidera approved in the European Union as a first-line oral treatment...
The European Commission has approved Tecfidera (dimethyl fumarate) as a first-line oral treatment for multiple sclerosis.
US dementia experts to share best practices in phase III clinical...
A US team of dementia experts will visit Russia to share experiences in phase III clinical research study.
BrainStorm signs definitive agreement with Mayo Clinic for ALS clinical trial
BrainStorm Cell Therapeutics has signed a definitive agreement with the Mayo Clinic to conduct Phase II of its ALS clinical trial.
Stenting without post-dilation is a safe and feasible strategy in the...
The results of a study published in the Journal of NeuroInterventional Surgery have shown that carotid stenting without post-dilation is safe and effective.
Myelin Repair Foundation and NIH enter into agreement to repurpose MRF-008...
The Myelin Repair Foundation has announced that it has entered a Cooperative Research and Development Agreement (CRADA) with the National Institutes of Health (NIH) to assess MRF-008, as a potential therapeutic for multiple sclerosis. The NIH are expect to instigate a clinical trial to assess this.
Canadian collaboration to fund anti-hypertensive drugs for Alzheimer’s disease research
A new funding collaboration to support a clinical trial investigating the potential for hypertension drugs to slow Alzheimer's disease progression has been announced between The Alzheimer's Drug Discovery Foundation of Canada and The W Garfield Weston Foundation.
Positive clinical performance of MS Precise for multiple sclerosis reported
New data has been reported supporting the clinical validation of MS Precise (DioGenix), a next-generation sequencing assay for the identification of patients with multiple sclerosis at first clinical presentation.
eNeura files 510(k) with the FDA for SpringTMS migraine treatment
eNeura has reported that it has filed a 510(k) with the US Food and Drug Administration (FDA) for the SpringTMS transcranial magnetic stimulation (TMS) device. SpringTMS is CE marked in Europe and available by prescription in the UK.
Brain surgeon teams with NASA scientists to develop 3D high-definition surgical...
Brain surgeon Hrayr Shahinian (the Skull Base Institute, Los Angeles, USA), a pioneer in minimally invasive surgery, and NASA's Jet Propulsion Laboratory in Pasadena, USA, have introduced the next generation of high-tech surgical instruments to remove tumours and treat other brain abnormalities.
Take head injuries seriously, says NICE
Time is of the essence when treating head injuries to avoid potentially serious complications, including disability or death, says the National Institute for Health and Care Excellence (NICE). Its updated guidance on the issue emphasises the importance of early detection and prompt treatment for both children and adults who have suffered trauma to the head.
Kessler Foundation neuroimaging study sheds light on mechanisms of cognitive fatigue...
The study, published in PlosOne, is the first study to use neuroimaging to investigate aspects of cognitive fatigue. The findings of the study support the part that the striato-thalamic-frontal cortical system plays in fatigue, suggesting a "fatigue-network" in multiple sclerosis.
Transcranial magnetic stimulation a “breakthrough treatment” for migraine patients
In NHS guidance, the National Institute for Health and Care Excellence (NICE) has announced the results from clinical trials investigating a portable transcranial magnetic device for the treatment of migraine.
NICE recommends Aubagio for multiple sclerosis
In final guidance, the National Institute for Health and Care Excellence (NICE) has recommended the use of Genzyme's multiple sclerosis drug teriflunomide (also called Aubagio) for adults with relapsing-remitting multiple sclerosis.
Middle-school girls continue to play football with concussion symptoms, study finds
Concussions are common among middle-school girls who play soccer, and most continue to play with symptoms, according to a study published in JAMA Pediatrics and by John W O' Kane, University of Washington Sports Medicine Clinic, Seattle, USA, and colleagues.
Vitamin D status associated with multiple sclerosis activity and progression
Vitamin D status appears to be associated with reduced disease activity in patients with multiple sclerosis and a slower rate of disease progression, according to a study by Alberto Ascherio, Harvard School of Public Health, Boston, USA, and colleagues.
Journal of the International Neuropsychological Society announces new editor-in-chief
Stephen M Rao has been named the new editor-in-chief of the Journal of the International Neuropsychological Society (JINS). Rao is the pioneer of the application of task-activated MRI and has applied this strategy to brain mapping for neurodegenerative conditions.
Richard Hammond backs children’s brain injury book launch
Top Gear presenter Richard Hammond is backing the launch of a book launch aimed at helping youngsters and their parents to better understand brain injuries and their hidden effects, it was reported in a press release from the Children's Trust.
Research confirms effectiveness of Cognigram, a new test for Alzheimer’s disease
A study from Australia, published online first in BMC Psychology, has confirmed the effectiveness a new test for Alzheimer's disease. The test is said to detect cognitive decline in patients by scoring attention/reaction and learning/memory and comparing the scores to traditional cognitive impairment indicators.
Stryker and NeuroLogica partner to provide surgical navigation with the BodyTom...
Stryker and NeuroLogica have announced an exclusive partnership agreement in which Stryker will promote and sell NeuroLogica's BodyTom portable full-body computed tomography (CT) scanner for the specialties of neurosurgery, spine surgery, orthopaedic surgery, and trauma surgery in conjunction with the Stryker NAV3i surgical navigation platform.
13,000 patients treated with Visius surgical theatre since 2005, study indicates
Results of an internal study of 40 worldwide Visius surgical theatre hospital customers indicates that nearly 13,000 patients have been treated using intraoperative MRI (iMRI) since the first installation in 2005.
National stroke summit tour to bring physicians across the USA together...
The Society of NeuroInterventional Surgery (SNIS) on 9 January has launched a 2014 national tour that will bring together physicians and medical personnel across the USA involved in stroke care to address current stroke treatment and the value of neurointerventional therapy in optimal stroke care delivery.
Next Wave brain stimulation for neuro-ophthalmologic patients available at German University
University Medical Center Hamburg-Eppendorf, Germany, is now offering the use of the EBS Next Wave brain stimulation device designed to expand the visual field of patients with impaired vision caused by optic nerve damage, it was announced by EBS Technologies.
Oscar 2 selected by PRA for pharmaceutical research trial for Alzheimer’s...
The SunTech Medical Oscar 2 ambulatory blood pressure monitoring system has been selected by PRA for use in a phase I clinical study. The study will investigate the inhibition of degenerative mechanisms to provide improved neuroprotection for those with Alzheimer's disease.
Device used to assess elevated brain pressure could benefit astronauts
A clinical research study of a non-invasive intracranial pressure measurement device has been launched by the National Space Biomedical Research Institute (NSBRI) and Vittamed, the developer of the device. The study will include the monitoring of neurological patients, as a prelude to possible future use with astronauts.
Apeiron signs regional license agreements for the commercialisation of a neuroblastoma...
Apeiron has announced the signing of two transactions as part of the worldwide commercialisation of APN311, an antibody-based immunotherapy in development for the treatment of children suffering from high-risk neuroblastoma.
Parkinson’s research study shows inosine compound raises urate levels
New findings published in JAMA Neurology from research into a potentially protective therapy for Parkinson's disease show that with medical supervision, urate levels may safely be raised while mitigating risk of complications such as gout. Previous research showed that higher levels of the antioxidant urate were associated with lower risk and slower progression of the disease.
BioCrea wins the Epilepsy Innovation Seal of Excellence Award
BioCrea announced on 9 January that the Epilepsy Foundation (USA) has presented the Epilepsy Innovation Seal of Excellence Award to BioCrea in recognition of the creation of the new anti-seizure platform, GABAA PAMs (LT GABAA PAM), to treat refractory epilepsy.
TauRx expands Alzheimer’s clinical trials in the USA
TauRx Therapeutics has expanded its international clinical trials by adding new research centres across the USA. The company says the addition of 35 clinical research centres means that more patients suffering from mild or moderate Alzheimer’s, and their caregivers, will have an opportunity to see if they qualify for participation in TauRx’s Alzheimer’s clinical trials.
Cognitive function in combat veterans with repeated brain injury examined
Researchers at Albert Einstein College of Medicine of Yeshiva University, New York, USA, in cooperation with Resurrecting Lives Foundation, are investigating the effect of repeated combat-related blast exposures on the brains of veterans.
First patient treated in Neuralstem China stem cell trial to treat...
The first patient was treated in the phase I/II NSI-566 stem cell trial to treat motor deficits from ischaemic stroke at BaYi Brain Hospital in Beijing, China, according to a press release.
Anxiety linked to higher long-term risk of stroke
A new study published in Stroke has identified that there is a higher risk of stroke in people with high levels of anxiety.
Implant allows rats with brain damage to rapidly recover motor skills
Scientists at the University of Kansas School of Medicine and Case Western Reserve University have developed a lightweight, battery-powered device that appears capable of repairing damaged pathways in the brain. The technology holds promise helping individuals who suffer from the damage left by stroke or head injuries.
Cerena TMS device for migraine headache pain gets FDA approval
The Cerena Transcranial Magnetic Stimulator (TMS) from eNeura Therapeutics is a prescription device used after the onset of pain associated with migraine headaches preceded by an aura.
Falls in Parkinson’s disease may be adversely impacted by deep brain...
New findings from the Parkinson Alliance show that falls are highly prevalent in Parkinson's disease and may be adversely impacted by deep brain stimulation therapy.
G8 countries agree to find a cure and treat dementia by...
Alzheimer’s Association president and CEO Harry Johns presented and participated in the first ever G8 Dementia Summit hosted by the UK. World leaders acknowledged that the Alzheimer’s and dementia crisis cannot be ignored. All of the G8 countries committed to the goal of identifying disease-modifying therapies or a cure for dementia by 2025.
Preliminary findings of phase IIa study of NurOwn reported as “extremely...
BrainStorm Cell Therapeutics has announced that the preliminary findings from the ongoing phase IIa dose-escalating trial evaluating NurOwn technology was presented at the 24th International Symposium on ALS/MND in Milan, Italy.
Clinical study of selective muscarinic M1 receptor agonist for Alzheimer’s disease...
Heptares Therapeutics has announced that it has initiated a phase I clinical study of HTL9936, the first fully-selective muscarinic M1 receptor agonist to enter clinical development. HTL9936 is an orally available, small molecule drug candidate using the Heptares GPCR structure-based drug-design (SBDD) platform.
US stroke deaths declining due to improved prevention and treatment
Stroke deaths in the USA have declined dramatically in recent decades due to improved treatment and prevention, according to a statement published in Stroke.
ALS Therapy Development Institute and Neurimmune partner to advance treatments for...
The ALS Therapy Development Institute (ALS TDI) and Anelixis Therapeutics have announced that it has formed a research partnership with Neurimmune to advance potential treatments for amyotrophic lateral sclerosis (ALS).
Thirty patients implanted in Advance study of deep brain stimulation for...
On 5 December, the company Functional Neuromodulation reported that 30 patients with mild Alzheimer's disease have been successfully implanted in the ongoing Advance study.
MRI Interventions and Imris technologies combine to lessen anxiety for deep...
Imris and MRI Interventions announced that Cook Children's Medical Center in Fort Worth, USA, is the first US paediatric hospital to offer asleep deep brain stimulation surgery to children suffering from dystonia by utilising the combination of MRI Interventions' ClearPoint Neuro Intervention System and an Imris Visius Surgical Theatre for real-time intraoperative image guidance and procedure visualisation.
First US implants of new Medtronic deep brain stimulation system
The first US implants of a novel deep brain stimulation system in research that is investigating the brain's response to the therapy has been announced by Medtronic.
Telemedicine can bring Parkinson’s care to “anyone, anywhere”
A new study shows that a neurologist in an office thousands of miles away can deliver effective specialised care to people with Parkinson's disease. For patients with the condition, these "virtual house calls" could allow them to live independently while effectively managing the symptoms of the disease.
Study of Prodigy neurostimulator to evaluate burst stimulation technology for chronic...
St Jude Medical has initiated a clinical study of the Prodigy neurostimulator, the first spinal cord stimulation system able to deliver a proprietary mode of stimulation therapy called burst stimulation. The study will evaluate whether burst stimulation can be more effective in managing chronic pain than traditional tonic stimulation.
Mechanical thrombectomy is beneficial if implemented by a collaborative neurointerventional team
A presentation at ESMINT (5-8 September, Nice, France) says that there are studies that indicate the benefit of mechanical thrombectomy if it is performed with a collaborative team of interventional neuroradiologists, neurologists and neurosurgeons and in dedicated stroke centres. Read more
Favourable results for real-world experience with the Precision Spectra Spinal Cord...
New retrospective data highlighting the Precision Spectra Spinal Cord Stimulator (Boston Scientific) System demonstrate the device provided highly significant pain relief three months after implantation. The results were presented at the North American Neuromodulation Society (NANS) 17th annual meeting in Las Vegas, USA (5-8 December).
Electrosonic stimulation as adjunctive therapy shows “significant improvements” in Parkinson’s disease...
A presentation on electrosonic stimulation as adjunctive therapy to dopaminergic treatments at the 5th International Symposium on Neuromodulation says that patients undergoing stimulation with the ESStim technology (Highland Instruments) saw significant improvements relative to baseline measurements when compared to sham stimulation in Parkinson's disease patients.
Novel rehabilitation device improves motor skills after stroke
Using a novel stroke rehabilitation device that converts an individual's thoughts to electrical impulses to move upper extremities has reported improvements in stroke patients' motor function and ability to perform day-to-day activities. Results of the study were presented at the annual meeting of the Radiological Society of North America (RSNA; 1-6 December, Chicago, USA).
Health Canada approves Aubagio for relapsing remitting multiple sclerosis
Genzyme has announced that Health Canada has approved Aubagio (teriflunomide) 14mg as monotherapy for the treatment of patients with relapsing remitting multiple sclerosis (RRMS) to reduce the frequency of clinical exacerbations and to delay the accumulation of physical disability.
Interim analysis of triple-blind, randomised iMRI trial proves better results for...
Fudan University (Shanghai, China) neurosurgeons have received a journal award at the Congress of Neurological Surgeons annual meeting for high-ranking clinical research using Visius Surgical Theatre for glioma tumours.
MR spectroscopy shows differences in the brains of preterm infants
Premature birth appears to trigger developmental processes in the white matter of the brain that could put children at higher risk of problems later in life, a study presented at the annual meeting of the Radiological Society of North America (RSNA; 1-6 December, Chicago, USA) says.
New Parkinson’s disease genes discovered
The Parkinson's Institute and Clinical Center, Sunnyvale, USA, says that they, in partnership with Population Diagnostics, have discovered a number of new genes relevant to the cause of Parkinson's disease.
Small series supports balloon microcatheter use for coil delivery in intracranial...
A study, published online first in the Journal of Neurointerventional Surgery has found that a single coaxial dual balloon microcatheter can be used to achieve coil placement and neck remodelling in selected oblong intracranial and cervical arterial aneurysms.
Vercise deep brain stimulation system receives CE mark approval for dystonia
The Vercise deep brain stimulation system (Boston Scientific) has received CE mark approval for the treatment of intractable primary and secondary dystonia.
Asahi Intecc launches new neurovascular guidewire in Japan
Ashai Intecc has announced the introduction and launch of an innovative neurovascular guidewire, Asahi Chikai black 18, in Japan. The company states that it plans to begin distributing the neurovascular guidewire in December.
Phase IIa study of RNS60 initiated for multiple sclerosis
Revalesio announced on 11 November a new collaboration with neurologist Roland Martin, head of Neuroimmunology and MS Research at the Department of Neurology, University Hospital Zurich, Switzerland. Martin is to conduct a phase IIa clinical trial of RNS60 in patients with relapsing-remitting multiple sclerosis (RRMS).
New brain technology to assist ALS patients will launch clinical trials
A new technology that will enable patients suffering from Amyotrophic lateral sclerosis (ALS)-also known as Lou Gehrig's Disease-to communicate via brain monitor, will hold its first clinical trials in Philadelphia, USA. The announcement was made at the BrainTech Israel conference in Tel Aviv (14-15 October).
Auto registration with surgical navigation from IMRIS and Stryker collaboration
IMRIS and Stryker have announced a non-exclusive collaboration of automatic image registration for spinal and cranial navigated procedures and VISIUS iCT and VISIUS iMRI surgical suites from IMRIS.
Sapiens appoints Jan Keltjens as president and CEO
Sapiens Steering Brain Stimulation (Sapiens) Advisory Board has appointed Jan C M Keltjens as the new president and CEO of the company, effective 21 October 2013.
Brainstem abnormalities found in SIDS infants, in all kinds of sleep...
Investigators at Boston Children's Hospital (Boston, USA) report that many infants dying suddenly and unexpectedly, in all kinds of sleep environments, have underlying brainstem abnormalities and are not all normal prior to death.
Walton Centre enters research partnership with Toshiba to develop brain scanning...
Toshiba Medical Systems has announced a research partnership with the Walton Centre (Liverpool, UK) to investigate new developments in neuroscience scanning applications and protocols. The research will be undertaken using Toshiba's Aquilion One CT scanning technology, acquired by the Walton Centre to provide data on a series of neuro applications.
Higher altitudes result in reduced concussion rates in high school related...
According to a recent study conducted at Cincinnati Children’s Hospital Medical Center, USA, high-school athletes who play collision sports at higher altitudes are less likely to suffer from concussions than those who play at lower altitudes.
Simulated cerebral anatomies could improve physicians’ training in neurointervention
Mentice announced on 15 November the release of the world's first solution to allow physicians to import medical-image data from cerebral anatomies of real patients into a simulated environment for the purpose of hands-on preparation and training.
Therapeutic Goods Administration approval for brain tumour visualisation drug
A novel drug which assists neurosurgeons to better visualise and remove malignant brain tumours has been approved by the Therapeutic Goods Administration (TGA).
A long walk may lower stroke risk in older men
Older men who walked at least one to two hours each day compared to less than half an hour per day had a reduced risk of stroke, a large population-based study reported in Stroke says.
RNS Stimulator gets FDA nod for device to treat epilepsy
The US Food and Drug Administration (FDA) have approved the RNS Stimulator (Neuropace) to help reduce the frequency of seizures in epilepsy patients who have not responded well to medications.
Large study finds 11 new genetic susceptibility factors for Alzheimer’s disease
The largest international study ever conducted on Alzheimer's disease, the International Genomics Alzheimer's Project (I-GAP) consortium has identified 11 new regions of the genome involved in the onset of this neurodegenerative disease.
High blood sugar makes Alzheimer’s plaque more toxic to the brain
The study supports growing evidence pointing to glucose levels and vascular damage as contributors to the progression of the deadly form of dementia.
Reverse Medical announces worldwide regulatory approvals and initial clinical experience with...
Reverse Medical has announced CE mark approval, FDA 510k clearance and initial clinical use of their Reverse microcatheter product line for intracranial neurovascular use.
Acandis gets CE mark approval for Derivo embolisation device
The Derivo device is intended for the treatment of patients with intracranial aneurysms who cannot be treated with current endovascular techniques or for patients who would have a high treatment risk with other endovascular or neurosurgical techniques.
New study suggests endovascular therapy may improve quality of life for...
According to recent research published in the September 2013 edition of Journal of Stroke and Cerebrovascular Diseases, patients see better results when they receive endovascular treatment at a dedicated stroke centre.
Asleep deep brain stimulation can relieve fear and anxiety surrounding surgery
Patients can now undergo asleep deep brain stimulation, under general anaesthesia, due to a revolutionary new platform called the ClearPoint Neuro Intervention System (MRI Interventions) that allows neurosurgeons to conduct the minimally invasive surgery inside an MRI scanner.
Depression was second leading cause of global disability burden in 2010
A study published this week in PLOS Medicine reports the most recent and comprehensive estimates on how much death and disability is attributable to depression, both worldwide and in individual countries and regions.
Measuring walking speed may paint picture of multiple sclerosis progression
Measuring the time it takes a person with multiple sclerosis to walk 25 feet may provide a clear picture of the progression of the disease, along with the severity of disability, according to a study published in the 30 October 2013 online issue of Neurology.
FDA approves second brain imaging drug to help evaluate patients for...
The US Food and Drug Administration has approved Vizamyl (flutemetamol F 18 injection), a radioactive diagnostic drug for use with positron emission tomography (PET) imaging of the brain in adults being evaluated for Alzheimer's disease and dementia.
In elderly, arterial stiffness linked to beta-amyloid plaques in brain
Even for elderly people with no signs of dementia, those with hardening of the arteries are more likely to also have the beta-amyloid brain plaques that are a hallmark of Alzheimer's disease, according to a study published in the 16 October 2013 online issue of Neurology.
Proprietary formulation of natural extracts reduces early cardiovascular events and deaths...
CHIMES is placebo-controlled trial of NeuroAiD. It has included 1,099 patients with ishaemic stroke who are receiving appropriate secondary prevention treatments. The frequency of early cardiovascular events and deaths during the three-month follow-up was halved in the NeuroAiD, group (p=0.025) without an increase in bleeding rate and non-vascular deaths.
Pulsar Vascular gets CE mark for Pulserider
Pulsar Vascular announced that it has received European CE mark approval for Pulserider. This unique implant is used to bridge the neck of cerebral aneurysms previously not amenable to endovascular therapy. This new device is implanted via standard, minimally invasive, endovascular techniques thus providing an alternative treatment option to open surgery.
Experts devise a way to cut radiation exposure in children needing...
A team of paediatric neurosurgeons and neuroradiologists at the Johns Hopkins Children's Center has developed a way to minimise dangerous radiation exposure in children with a condition that requires repeat CT scans of the brain. The experts say they reduced exposure without sacrificing the diagnostic accuracy of the images or compromising treatment decisions.
Pitavastatin could help prevent ischaemic complications with carotid artery stenting, EPOCH-CAS...
Data presented at CIRSE (14-18 September, Barcelona, Spain) by a team of investigators from Japan showed that pre-treatment with pitavastatin significantly reduced the frequency of periprocedural ischaemic complications associated with carotid artery stenting.
“Gold standard” imaging study reveals no association between venous narrowing and...
A study that used catheter venography has found that there is no significant difference between rates of venous narrowing in people with multiple sclerosis, their unaffected siblings, and unrelated people without the neurological condition.
ImThera Medical completes enrolment in multicentre neurostimulation study for obstructive sleep...
ImThera Medical has announced that the final patient has been implanted in its Targeted hypoglossal neurostimulation #2 (THN2) study. The THN2 Study is an international, prospective multicentre study that will evaluate the safety and efficacy of the Aura6000 sleep therapy system for the treatment of moderate to severe obstructive sleep apnoea.
Neuros Medical gets US FDA regulatory approval for Altius
Neuros Medical has announced that it has received an investigational device exemption (IDE) approval from the US Food and Drug Administration for their implantable generator. The Altius device delivers patented high frequency Electrical Nerve Block technology for patients suffering from chronic pain.
No clear recommendation for thrombectomy
Some of the most important trends in stroke therapy involve further developments in the area of thrombolytic therapy as well as new insights into the mechanical removal of blood clots in acute strokes through minimally invasive catheter procedures, according to Werner Hacke, University of Heidelberg, and vice president of the World Federation of Neurology, at the XXI World Congress of Neurology (WCN, Vienna, 21-26 September 2013).
Drug developed in Cambridge approved for treatment of multiple sclerosis
A new treatment for multiple sclerosis was approved recently by the European Medicines Agency (EMA). The agency has approved the drug Alemtuzumab, to be known by the brand name Lemtrada.
New way to track stem cell migration could advance treatments for...
A new study in the September issue of Stem Cells Translational Medicine shows how a team of scientists led by Rex Moats, and Karen Aboody, have discovered a safe and effective way to track the migration and distribution of neural stem cells used in the treatment of invasive brain tumours.
Initial experience shows ADAPT is a simple and effective technique to...
The development of new revascularisation devices has improved recanalisation rates and time but not clinical outcomes. "The technique with which new stroke thrombectomy devices are used may be as important as the device itself," say Aquilla Turk, Charleston, USA, and colleagues.
First patient enrolled in SCAFFOLD clinical study for carotid stenting
Multicentre investigational study begins at the Medical University of South Carolina, Charleston, USA, to evaluate the safety and efficacy of Gore's carotid stent by comparing it to a performance goal developed from carotid endarterectomy outcomes.
Low complication rates and good clinical outcome with Y-stent coiling for...
A retrospective, multicentre series, published in Neurosurgery in September, demonstrates that stent-assisted coiling with two stents in a Y configuration has a low rate of complications and a low incidence of re-treatment.
Codman announces European launch of next generation Revive SE for acute...
The new version of the Revive SE thrombectomy device offers enhanced navigation through the cerebral vasculature and rapid restoration of blood flow to the brain after an ischaemic stroke.
Neurosurgeon performs first deep brain stimulation with Renaissance to treat Parkinson’s...
On 19 August 2013, Nizam Razack performed the world's first deep brain stimulation using the Renaissance Guidance System (Mazor Robotics) at Celebration Health hospital in Orlando, USA. Razack then performed the same procedure on two more patients with positive results.
Use of balloon guide catheter with Solitaire improves results of acute...
A substudy from the NASA registry suggests that the use of a balloon guide catheter with the Solitaire Stent retriever in acute ischaemic stroke results in superior recanalisation results, faster procedure time, decreased need for rescue therapy, and improved clinical outcome.
Focused ultrasound thalamotomy works to reduce essential tremor
The results of a pilot study, published in the New England Journal of Medicine, show that delivery of high-intensity focused ultrasound through the intact human cranium with magnetic resonance imaging (MRI) guidance reduced tremor by creating a small ablation deep in the brain
In mild strokes, ultra-early treatment may eliminate risk of disability
In the case of mild or moderate strokes, getting treatment ultra-fast (within 90 minutes of experiencing symptoms) reduces the risk of suffering disability, according to a new study reported in the American Heart Association's journal, Stroke.
First laser ablation for brain tumour performed using ClearPoint neurointervention system...
Joint application of the two enabling medical technologies can exhibit benefits of intraoperative MRI for direct visualisation when navigating to treat brain tumours and neurological disorders.
Robot treats brain clots with steerable needles
Surgery to relieve the damaging pressure caused by haemorrhaging in the brain is a perfect job for a robot. A new image-guided surgical system under development at Vanderbilt University, Nashville, USA, has the robot employ steerable needles (about the size of those used for biopsies to penetrate the brain with minimal damage) and suction away the blood clot that has formed.
Alzheimer’s and Parkinson’s do not appear to share common genetic risk,...
A study by Valentina Moskvina, Cardiff University School of Medicine, Wales, United Kingdom, and colleagues, examined the genetic overlap between Parkinson's and Alzheimer's. The study was
published online on 5 August in JAMA Neurology.
Medtronic introduces first MRI-safe neurostimulation system for chronic pain
With the first US implants of its new RestoreSensor SureScan MRI neurostimulation systems, Medtronic has introduced an implantable system for use in the treatment of chronic, intractable back and/or limb pain that is approved by the US FDA as conditionally safe for full-body MRI.
Offering neuromodulation earlier could be better in complex regional pain syndrome
Spinal cord stimulation should be considered earlier, rather than a last resort for treating a rare but debilitating chronic pain condition known as complex regional pain syndrome, according to a research analysis in Neuromodulation: Technology at the Neural Interface, the journal of the International Neuromodulation Society.
FIRST trial provides real-world data on natural evolution of ischaemic stroke...
The FIRST trial, a prospective, multicentre study sponsored by Penumbra, provides data to assess the natural history of stroke in a population of patients who, circumstances notwithstanding, would have otherwise been treated with intra-arterial therapy.
Endovascular sonolysis results in “safe and potentially effective” revascularisation in acute...
A small, prospective study, published in the July issue of American Journal of Neuroradiology, showed that sonolysis using the EkoSonic Endovascular system resulted in safe and potentially effective revascularisation in patients experiencing acute ischaemic stroke.
Growing brain aneurysms most likely to rupture
Cerebral aneurysms of all sizes, even small ones below seven millimetres, are 12 times more likely to rupture if they are growing, according to a new study published online in the journal Radiology.
New US registry planned for spectrum of neurovascular conditions
The Society of Neurointerventional Surgery (SNIS) will host a new national registry, entitled the Neurovascular Quality Initiative (NVQI), which will make it possible to measure performance and outcome data across a spectrum of neurovascular conditions, including stroke and aneurysms.
Continuous cell loss in parahippocampal gyrus could signal later diagnosis of...
Early signs of Alzheimer's disease can be detected years before diagnosis, according to researchers at Birmingham City University, Birmingham, UK.
FDA clearance for AIGISRx Neuro antibacterial envelope
TYRX has announced FDA approval of the AIGISRx antibacterial envelope for use with neurosurgery patients undergoing vagus nerve stimulation for seizure disorders and depression.
Microvention enrols first patient in US clinical trial to test new...
Microvention has announced the first successful enrolment of a patient in a multicentre, prospective, pivotal US clinical trial to demonstrate the safety and efficacy of its Fred flow diversion system in the treatment of intracranial aneurysms.
Boston Scientific launches trial to evaluate neurostimulation for treatment of chronic...
The first patient has been treated in the OPTIMISE clinical trial evaluating safety, efficacy of occipital nerve stimulation using the Precision system, a company press release finds.
Lowering systolic blood pressure probably benefits lacunar stroke patients, SPS3 finds
Lowering systolic blood pressure to <130mmHg in patients with recent lacunar stroke significantly reduced intracerebral haemorrhage, and is likely to reduce all recurrent stroke. These findings from the SPS3 (Secondary prevention of small subcortical strokes) trial were presented at the European Stroke Conference and published simultaneously online in The Lancet.
New estimates suggest that prevalence of dementia is falling in UK
The proportion of people with dementia in the UK has fallen, according to new estimates from the first ever study to show how dementia prevalence has changed in the UK population over time. The study was published in The Lancet.
Study finds factors that may cause fluctuations in deep brain stimulation...
Deep brain stimulation therapy blocks or modulates electrical signals in the brain to improve symptoms in patients suffering from movement disorders such as Parkinson's disease, essential tremor and dystonia, but a new study has suggested that several factors may cause electrical current to vary over time.
Stroke recovery theories challenged by new studies looking at brain lesions,...
Stroke survivors left weakened or partially paralysed may be able to regain more arm and hand movement than their doctors realise, experts at The Ohio State University Wexner Medical Center, Columbus, USA, who have published two new studies evaluating stroke outcomes, have said.
Penumbra launches 5Max-Ace to treat acute ischaemic stroke
On 8 July, Penumbra announced the launch and wide availability of the 5Max Ace, a next generation clot extraction device that uses aspiration alone to engage and remove blood clots that cause an acute ischaemic stroke.
Biogen Idec submits application for approval of Plegridy in multiple sclerosis
Biogen Idec announced that it has submitted a Biologics License Application to the US Food and Drug Administration (FDA) for approval of Plegridy (peginterferon beta-1a), the company's pegylated subcutaneous injectable candidate for relapsing forms of multiple sclerosis.
Coiling of unruptured intracranial aneurysms can relieve headaches in elderly patients
A study from China found that endovascular coiling of unruptured intracranial aneurysms succeeded in relieving most patients from headache in a small cohort of elderly patients. The study was published in the American Journal of Neuroradiology in June.
Neurosurgeons verify eye tumour drug delivery localisation with iMRI in VISIUS...
IMRIS, on the 19 June 2013 announced that Yale-New Haven Hospital, New Haven, USA, neurosurgeons have confirmed effective chemotherapy delivery in treating eye cancer using intraoperative imaging in the VISIUS Surgical Theatre.
Deep brain stimulation system demonstrates improvement in motor function in Parkinson’s...
The VANTAGE study interim data presented at International Congress of Parkinson's Disease and Movement Disorders in Sydney, Australia, reported that using the Vercise deep brain stimulation system (Boston Scientific) saw improvement in motor scores in Parkinson's disease patients.
Pipeline embolization device is safe for treatment of large and giant...
Results from a study showed that the Pipeline device offered safe and effective treatment of large and giant aneurysms.
CLOTS 3 study shows sequential compression sleeves reduce deep vein thrombosis...
Intermittent pneumatic compression, in which thigh-length sleeves are used to compress the veins in the legs, mimicking the effects of walking, is an effective method of reducing the risk of deep vein thrombosis and possibly improving survival patients who are immobile after stroke, the study reveals.
ADvance study implants 20 patients to examine deep brain stimulation for...
On 12 June 2013, Functional Neuromodulation announced that the ADvance study has implanted 20 mild Alzheimer's patients with deep brain stimulation systems, and the US Food and Drug Administration (FDA) has approved expansion of the study from 20 to 30 US patients in combination with 20 subjects approved in Canada.
Thomson Reuters and One Mind for Research collaborate to help disseminate...
The collaboration of Thomson and One Mind has seen the transmitting of central nervous system and traumatic brain injuries through tranSMART-a data sharing and analytics platform.
Deep brain stimulation in treatment-resistant obesity linked to weight loss
Trial results, presented at the International Neuromodulation Society's World Congress, has proven that, deep brain stimulation set at an optimal setting to increase metabolism can help morbidly obese patients lose weight.
CE mark for REVIVE SE thrombectomy device for clot removal in...
Codman Neuro has announced at the Live Interventional Neuroradiology and Neurosurgery Course (LINNC) in Paris, France, that it has obtained CE marking for the REVIVE SE, a self-expanding clot removal device for use in treating acute ischaemic stroke.
International guidance on using neurostimulation for chronic pain formulated at INS
It was announced at the World Congress of the International Neuromodulation Society (INS) in Berlin, Germany, that the Neuromodulation Appropriateness Consensus Committee has issued an international guidance on implementing neurostimulation for chronic pain.
Frequent football ‘heading’ may lead to brain injury
Researchers at Albert Einstein College of Medicine, Yeshiva University, New York, USA, have shown, according to a press release, that football players who frequently head the ball have brain abnormalities resembling those found in patients with concussion (mild traumatic brain injury).
Integrated lead-to-pulse generator interconnect system for implantable neurostimulators launched
Evergreen Medical Technologies has announced the launch of the Encompass Lead-Interconnect System, which is expected to be featured at the International Neuromodulation Society World Congress. The system is approved for use with implantable neurostimulator pulse generator devices.
NeuroVision eye test trialled as a screening tool for Alzheimer’s disease
According to a press release, researchers in Perth, Australia, are trialling a NeuroVision eye test as a screening tool for Alzheimer's disease in conjunction with MRI and PET imaging.
Results of pilot study for high frequency electrical nerve block technology...
An upcoming presentation at the International Neuromodulation Society 11th World Congress (8-13 June, Berlin, Germany) is expected to detail the results of a study that evaluated a high frequency electrical nerve block technology (Neuros Medical) for the treatment of chronic pain.
MRI Interventions, IMRIS and Tocagen combine their technologies for drug delivery...
The first combination of the ClearPoint neurointerventional system within the VISIUS surgical theatre has reported to have achieved real-time MRI-guided delivery of an investigational gene therapy drug-Toca 511.
CE mark for Sequent Medical’s Web SL family of neurovascular aneurysm...
Sequent Medical announced that it has received CE Mark approval for its Single-Layer family of Web aneurysm embolisation devices and has commenced a controlled release of these new models in select neurovascular centres in Europe, according to the company.
CE mark approval for the first deep brain stimulation visualisation system
Boston Scientific has announced the CE mark for the use of the Guide deep brain stimulation visualisation system. The system is available in Europe and is only for investigational use in the USA.
Israel Brain Technologies announces Israel’s first international brain technology conference
Israel Brain Technologies has announced the first BrainTech Israel 2013 conference which is intended to showcase new opportunities and next generation brain technologies.
Integra LifeSciences debuts Camino Platform at AANS
Integra LifeSciences announced the debut of its next generation of the Camino platform, an advanced monitoring system for continuous intracranial pressure monitoring and cerebral spinal fluid drainage.
Black women have a higher incidence of multiple sclerosis than white...
Findings from research into the incidence of multiple sclerosis at Kaiser Permanente, Oakland, USA, dispute the previously common belief that black women are at a lower risk of developing the disease.
UCLA perform new transcarotid stenting with dynamic flow reversal procedure
Physicians at UCLA were, according to a release, first to perform a new procedure on west coast of America to safely open blocked carotid arteries as part of a clinical trial for examining high surgical risk patients.
Promising results for clinical trial for stem cell-based treatment of ALS...
Results of the first American clinical trial for stem cell-based treated for amyotrophic lateral sclerosis (ALS) were presented at the Annual Scientific Meeting of the American Association of Neurological Surgeons (AANS), 27 April to 1 May 2013, New Orleans, USA.
Benefits of intra-operative neuromonitoring might not outweigh the costs
Researchers announced findings during the 81st American Association of Neurological Surgeons (AANS) Annual Scientific Meeting (27 April-1 May 2013, New Orleans, USA) regarding the value of intra-operative neuromonitoring when comparing its use in influencing patient outcomes compared to the added cost it incurs.
Phase I study of transcranial MR-guided focused ultrasound thalamotomy sees reduced...
In a presentation at the American Association of Neurological Surgeons (AANS) Annual Scientific Meeting (27 April-1 May 2013, New Orleans, USA), William Jeffery Elias spoke about the results of a trial for the use of transcranial MR-guided focused ultrasound thalamotomy for essential tremor. The results showed a significant improvement.
Study demonstrates significant pain reduction with Dfine Star tumour ablation system...
The Dfine Star ablation system represents a safe and effective new technique for the treatment of painful malignant spinal metastases, according to a new study.
Medtronic announces FDA Class I recall of deep brain stimulation lead...
Medtronic has issued an Urgent Medical Device Correction notification to advise physicians the use of the lead cap as part of the the Medtronic deep brain stimulation lead kits can lead to damage.
Implanted device predicts epilepsy seizures in humans
A study, published in The Lancet Neurology has reported on an implanted device in the brain that predicts the onset of procedures in epilepsy patients.
IMRIS to exhibit VISIUS iCT imaging solution at AANS
IMRIS is expected to exhibit novel VISIUS iCT imaging solution at the American Association of Neurological Surgeons (AANS) Annual Meeting, 27 April to 1 May 2013, New Orleans, USA.
Boston Scientific launches Precision Spectra Spinal Cord Stimulator System in the...
On the 12 April 2013 Boston Scientific received approval from the US Food and Drug Administration (FDA) and has begun a limited launch of the Precision Spectra Spinal Cord Stimulator System.
St Jude Medical receives CE mark for deep brain stimulation systems...
On 10 April 2013, St Jude Medical announced European CE mark approval of its Brio, Libra and LibraXP deep brain stimulation systems for managing the symptoms of intractable primary and secondary dystonia.
Monteris Medical’s NeuroBlate System receives 510(K) clearance
Monteris Medical announced that the US Food and Drug Administration (FDA) have issued a second 510(k) clearance for their magnetic resonance imaging (MRI)-guided ablation device for brain tumours and other lesions.
Medtronic initates US trial to investigate the use of peripheral nerve...
Medtronic announced the first patient enrolments in the SubQStim II pivotal clinical trial to pursue US Food and Drug Administration (FDA) approval of peripheral nerve stimulation, also known as subcutaneous nerve stimulation, for the reduction of chronic, intractable post surgical back pain.
Gamma Knife Perfexion system used at Sacred Heart Health System to...
American Shared Hospital Services a provider of turn key technology solutions for advanced radiosurgical and radiation therapy services, announced today that the Gamma Knife Perfexion system has been supplied to Sacred Heart Health System in Florida, USA, and has been used to treat patients.
The Second Annual UAE Epilepsy Congress to be held in...
Abu Dhabi will host the Second Annual UAE Epilepsy Congress (26-27 April 2013). The congress is hosted under the umbrella of the Emirates League Against Epilepsy (ELAE).
The American Academy of Neurology launches app for identifying concussion in...
At the American Academy of Neurology's Annual Meeting in San Diego, USA, the society launched their new app-Concussion QuickCheck-to help identify concussion for people who have sustained a head injury during sport, or sports injury which may lead to concussion.
The American Academy of Neurology issues updated sports concussion guidelines
Athletes with suspected concussion should be removed from play, according to the American Academy of Neurology's new guidelines that were issued on 18 March 2013.
InspireMD receives CE mark for Carotid Embolic Protection Stent
InspireMD's Carotid Embolic Protection Stent, based on InspireMD's proprietary MicroNet mesh technology, is designed to provide procedural and post-procedural distal embolic protection in carotid artery stenting procedures and reduce risk of stroke events, according to the company.
Early detection of natalizumab treatment for multiple sclerosis complication may improve...
A study, presented at the American Academy of Neurology's Annual Meeting (16-23 March, San Diego, USA) suggested that natalizumab for the treatment of multiple sclerosis increases the risk of progressive multifocal leukoencephalopathy (PML). The study author said that early detection of PML can help mitigate the effects.
Philips aims to screen one million people for obstructive sleep apnoea...
According to Philips, snoring is a key sign of sleep apnoea which can lead to serious chronic diseases such as diabetes or heart disease.
Study suggests possible marker of Alzheimer’s disease associated with worse sleep...
Yo-El S Ju, Washington University School of Medicine, St Louis, and colleagues suggested in a study published online first in JAMA Neurology, that amyloid deposition in the preclinical stage of Alzheimer's disease appears to be associated with worse sleep quality but not with changes in sleep quantity.
Brain imaging after mild head injury or concussion can show lesions...
A study, intended to be presented at the American Academy of Neurology's Annual Meeting (16-23 March 2013, San Diego, USA), suggested that after post mild traumatic brain injury, imaging can show linear and microbleed lesions often associated with more traumatic brain injury.
InVivo Therapeutics submits updated IDE to begin spinal cord injury human...
InVivo applied for Investigational Device Exemption (IDE) to begin human studies of its biopolymer scaffolding for the treatment of spinal cord injury and other neurotrauma. InVivo reported that they will also be seeking Humanitarian Use Device (HUD) status.
Microvention announces the opening of neurovascular manufacturing facility in Costa Rica
Microvention opened its neurovascular facility in San Jose, Costa Rica. According to the company, it is the first specialist neurovascular facility in Costa Rica.
Study examines cerebrospinal fluid markers in relation to Alzheimer’s disease
A study, led by Maureen Handoko, University of Minnesota, Minneapolis, USA, investigated the cerebrospinal fluid of older patients at risk of Alzheimer's disease vs. younger normal controls in order to assess the relationship of Alzheimer's with tau and cerebrospinal fluid.
Deep brain stimulation could help treat severely anorexic patients who have...
Scientists, in a pilot study, have used deep brain stimulation to treat six patients with severe and enduring anorexia who had not responded to any existing treatments, according to new research published in The Lancet.
Research funded by the ALS Association found gene mutations can cause...
Published in Nature and funded by the ALS Association, a study said that mutations in genes for certain RNA-binding proteins can cause ALS and brain, muscle, and bone diseases. Researchers suggested that prevention of protein aggregation may be a therapeutic option for the treatment of ALS.
NeuroDerm phase I study of ND0612 reports positive results for the...
According to NeuroDerm, ND0612, in its first phase I trial has reported positive results in the reduction of motor complications in Parkinson's disease patients as it maintains a steady state concentration of levodpa plasma levels.
Study suggests lower extremity functional electrical stimulation cycling can promote neurological...
It was suggested by Kennedy Krieger's International Center for Spinal Cord Injury, Baltimore, USA, that activity-based restorative therapy programmes may provide substantial benefits, including neurological benefits, for persons with chronic spinal cord injury from a study conducted by the centre.
FDA Advisory Panel recommends the approval of the NeuroPace RNS System...
The NeuroPace RNS System, according to the company, is a novel device which use responsive brain stimulation to treat patients who do not respond to medication and has been recommended for FDA approval following a panel which voted its benefits outweigh the risks of use.
Advice from NICE aims to improve commissioning of care for people...
The National Institute for Health and Clinical Excellence (NICE) on the 28 February 2013 issued an updated guide for commissioners to support the integrated commissioning of high-quality, evidence-based services for the diagnosis and management of the epilepsies in adults, children and young people.
New quality standards for the epilepsies in adults, young people and...
According to a release, NICE quality standards describe high-priority areas for quality improvement in a defined care or service area for the treatment of epilepsy. They are derived either from NICE guidance or NICE accredited sources, and apply right across the NHS in the UK.
High-frequency spinal cord stimulation appears to be efficacious in chronic back...
Jean-Pierre van Buyten and colleagues assessed the efficacy of the Senza rechargeable high-frequency spinal cord stimulation system for the treatment of patients with chronic, intractable back and leg pain who have previously failed treatment with conventional stimulation.
NIH-funded study confirms consistently good acute stroke outcomes with Penumbra System
At the International Stroke Conference, Honolulu, Hawaii, (6-8 February 2013) data were presented from the IMS III trial comparing two different treatment approaches: combined intravenous and intra-arterial therapy, and standard intravenous r-tissue plasminogen activator (r-tPA), to restore blood flow to the brain.
ElectroCore receives Australian and Colombian regulatory approval for GammaCore Vagal Nerve...
GammaCore therapy has been approved for commercial sale by the Therapeutic Goods Administration in Australia and the National Institute of Surveillance of Medicine and Foods in the Ministry of Health in Colombia. GammaCore's non-invasive, non-pharmaceutical, neuromodulation therapy is indicated for the acute and/or prophylactic treatment of primary headache.
InSightec’s ExAblate Neuro receives the FDA approval for its pivotal phase...
InSightec, with FDA approval, will be assessing the safety and effectiveness of the ExAblate Neuro for treatment of patients with essential tremor in a phase III trial. The ExAblate Neuro received the CE mark in Decemeber 2012.
New research indicates use of Medtronic deep brain stimulation therapy is...
EARLYSTIM study results published in The New England Journal of Medicine show improvements in patient quality of life and motor disability.
Fukuda Denshi to distribute Boston Scientific Neuromodulation devices in Japan
Boston Scientific signed an agreement on 20 February 2013 with Fukunda Denshi to distribute the Boston Scientific Spinal Cord Precision Plus System in Japan for the treatment of chronic neuropathic pain.
Study shows continued improvement in patients’ neuropathic pain scores after deep...
A study, published in Neurosurgery has reported on the long term outcomes of difficult-to-treat neuropathic pain treated with deep brain stimulation.
Cooling the brain may prevent trauma-induced epilepsy
A new study in rats suggests that gently cooling the brain after injury may prevent patients who often experience epileptic seizures that are difficult to control in weeks, months and years after severe head injury. The researchers reported their findings in Annals of Neurology.
Penumbra launches 5MAX DDC and 4MAX DDC Distal Delivery Catheters
Penumbra launches at the 3rd Society of NeuroInterventional Surgery (SNIS) International Endovascular Stroke Conference and Joint Cerebrovascular Section Annual Meeting the new DDC family intended to aid in the delivery of a broad array of neurovascular therapies.
University of Tsukuba Hospital neurosurgeons perform initial cases using recently installed...
An initial case of tumour removal has been performed by neurosurgeons at the University of Tsykuba Hospital, Japan, using the VISIUS Surgical Theatre.
A novel balloon technique is safe during endovascular intervention for vertebral...
Vikram Huded and colleagues have reported on the use of a novel technique for stenting the vertebral artery in the presence of a thrombus.
Study says that there is no evidence to support concerns that...
A study, led by David Irwin, has said that recipients of a cadaver-derived growth hormone did not appear to be at an increased risk of Alzheimer's or Parkinson's disease.
Research presented at ISET shows controversial multiple sclerosis treatment may ease...
Angioplasty to treat multiple sclerosis, according to a presentation at ISET, may improve symptoms however, there is a lack in data to suggest whether this is correct said Marco Magnano.
Brain stimulation in conjunction with antidepressants may help depression, study says
Researchers in the USA and Brazil have said that transcranial direct current stimulation with sertaline may help ease depression symptoms.
Zhengzhou neurosurgeons perform first iMRI case in VISIUS Surgical Theatre in...
IMRIS has announced that neurosurgeons at the First Affiliated Hospital of Zhengzhou University in Zhengzhou City, China, have successfully performed the first case involving an adult male brain tumour patient using the recently installed VISIUS Surgical Theatre.
Nevro announces publication of six-month clinical data for Senza HF10 High-Frequency...
Six-month data from Europe has provided evidence that high-frequency spinal cord stimulation therapy is safe, efficacious and provides paresthesia free pain relief in a difficult-to-treat population.
PROMISE study to investigate neurostimulation for failed back surgery syndrome launched...
The first patients in the PROMISE study have been treated with spinal cord stimulation with optimal medical management vs. optimal medical management only for failed back surgery syndrome.
Recall of Vycor Viewsite Brain Access System due to unidentified fibre...
Vycor Medical has recalled its Vycor Viewsite Brain Access System because an unidentified black fibre was found on the device.
Medical societies unite on patient-centered measures for nonsurgical stroke interventions
The Society of Interventional Radiology has published guidelines that capture consensus on clinical practice of intra-arterial stroke revascularisation.
The American Heart Association/American Stroke Association launches the Together to End...
The American Heart Association/American Stroke Association is to launch the Together to End Stroke to help people recognise the symptoms of stroke.
Neurostimulation from Medtronic receives CE mark for use with full-body MRI...
Medtronic has introduced in Europe their first implantable neurostimulation systems indicated for use in the treatment of chronic back and/or leg pain that are designed for full-body Magnetic Resonance Imaging (MRI) scans under specific conditions.
Samsung Electronics America acquires NeuroLogica
Samsung's acquisition of NeuroLogica is expected to strengthen Samsung's growing medical equipment business, according to a company release.
New extremely soft bare platinum coil is a safe and effective...
Kei Harada, Department of Neurovascular Surgery, Fukuoka Wajiro Hospital, Japan, and others have reported the first use in their centre of an extremely soft platinum coil for the treatment of cerebral aneurysms.
Preoperative Onyx embolization of hypervascular head, neck, and spinal tumours is...
"Onyx embolization was effective as it precipitates gradually in a radial fashion, forming a cast. The slow precipitation property allows a more slow and precise injection, allowing deeper penetration into the tumour capillary bed," according to a study on embolization for head, neck and spinal tumours.
Mild traumatic brain injuries could be associated with cognitive impairment in...
A study by John Hart and others has said that mild traumatic brain injuries in athletes is associated with white matter disruption and cerebral blood flow changes which could be detected using MRI.
Functional Neuromodulation appoints Vince Owens to its board of directors
Vince Owens, previously of Intelect Medical, has joined Functional Neuromodulation's-a deep brain stimulation company-board of directors.
Depressed post-stroke patients have a higher risk of death than their...
A US study has found that patients who are depressed after suffering a stroke are four times more likely to die than stroke survivors who are not depressed.
SanBio announces enrolment of the second cohort of patients in its...
A clinical trial is testing the safety and efficacy of a novel allogeneic stem cell therapy product, SB623 for chronic stroke patients.
Transbrachial carotid artery stenting with novel sheath guide is feasible and...
Takahisa Mori, Shonan Kamakura General Hospital Stroke Center, Kamakura City, Kanagawa, Japan, and colleagues reported on their first experience of transbrachial carotid artery stenting using a novel sheath guide in the Journal of NeuroInterventional Surgery.
Internet-based survey shows patterns of variability in neurointerventional practice for acute...
A study, published in the Journal of NeuroInterventional Surgery and conducted by Brijesh P Mehta, Boston, USA, and colleagues has assessed the variability in neurointerventional practice for intra-arterial therapy with tPA for patients with major acute ischaemic stroke secondary to large vessel occlusions.
Covidien opens patient enrolment for SWIFT PRIME acute ischaemic stroke study
A new clinical trial from Covidien aims to examine the effectiveness of advanced mechanical thrombectomy in the treatment of stroke.
Phenox receives CE mark approval for pCONus Bifurcation Aneurysm Implant
The pCONus implant for treatment of complex, wide neck intracranial bifurcation aneurysms has been granted the CE mark for commericalisation in Europe.
New development at BrainStorm for commercialisation of NurOwn
BrainStorm has received a development in commericalising NurOwn for stem cell therapy for the treatment of amyotrophic lateral sclerosis.
Asahi Intecc to launch neurovascular guidewires in USA and Europe
Asahi Intecc has announced that it will start the commercial distribution of its neurovascular guidewires (Asahi Chikai and Asahi Chikai 10) in the United States and Europe in January 2013.
Managing atrial fibrillation may prevent the risk of stroke
In a presentation at the UK Stroke Forum, Andre Ng told delegates about using drug treatments and devices to manage atrial fibrillation for stroke prevention.
Late-life depression associated with prevalent mild cognitive impairment and an increased...
A study, led by Edo Richard, has found depression was related to a higher risk of prevalent mild cognitive impairment and dementia.
Accordion effect and spindle shape deformation can affect high-porosity and flow-diverting...
A study investigating high-porosity and flow-diverting stent deformities in canine intracranial aneurysms has found deformities occurred in vivo that did not occur in in vitro (glass tube) testing.
Solitaire FR revascularisation device receives regulatory approval in Canada
The Solitaire FR is used in the retrieval of blood clots in stroke patients and has been approved by Health Canada.
Intra-arterial revascularisation with stents safe and effective for stroke patients with...
Some stroke patients may benefit from cerebral angioplasty and stent placement, according to a new study published online ahead-of-print in the 11 December issue of the journal Radiology.
Researchers discover new links leading to neuron death
The discovery, funded by The ALS Association, highlights the links between several proteins known to go awry in ALS and a related disease, frontotemporal dementia.
ADvance study of deep brain stimulation for Alzheimer’s disease performs first...
ADvance will evaluate the safety and potential clinical benefit of deep brain stimulation of the fornix (DBS-f), a major inflow and output pathway in the brain's memory circuit, for patients with mild Alzheimer's.
ExAblate Neuro transcranial focused ultrasound system gets the CE mark for...
ExAblate Neuro (InSightec) has been awarded the CE mark for the treatment of neurological disorders including: essential tremor, Parkinson's disease and neuropathic pain.
Toshiba launches Aquilion One Vision Edition CT scanner
The CT scanner is specially designed to detect cardiovascular and neurovascular diseases.
Stroke treatment performed by interventional radiologists in collaboration with diagnostic neuroradiologists...
Lars Fjetland, Stavanger, Norway, and others investigated the possibility of collaboration between vascular interventional radiologists, diagnostic neuroradiologists and stroke neurologists for neurointerventional treatment of acute stroke as a strategy for centres lacking of specialised interventional neuroradiologists.
Active lifestyle boosts brain structure and slows Alzheimer’s disease
An active lifestyle helps preserve gray matter in the brains of older adults and could reduce the burden of dementia and Alzheimer's disease, according to a study presented at the annual meeting of the Radiological Society of North America (RSNA).
Researchers find evidence that brain compensates after traumatic injury
A new study has found that diffusion tensor imaging may be able to predict which patients who have experienced concussions will improve.
Worldwide incidence of traumatic brain injury could be six times higher...
The New Zealand population-based study, published online first in The Lancet Neurology, found that rates of traumatic brain injury (TBI) and particularly mild TBI were far higher than in other high-income countries in Europe and North America.
National Institute for Health and Clinical Excellence recommends BIS Brain Monitoring...
The BIS Brain Monitoring System (Covidien) helps clinicians assess patient consciousness levels through electrical activity in the brain.
Perispinal administration of etanercept helps to improve neurological dysfunction following stroke...
An observational study has provided clinical evidence that chronic neurological dysfunction from stroke or traumatic brain injury can rapidly improve following perispinal administration of etanercept, a drug that targets brain inflammation, even years or decades after the initial event.
Worse clinical outcomes in stroke patients with difficult catheter access in...
Marc Ribo, Barcelona, Spain, and colleagues, in a study published ahead-of-print in the Journal of Neurointerventional Surgery, said that difficult catheter access to the carotid artery during endovascular treatment of stroke patients is common and is associated with worse clinical outcomes.
New balloon-stent technique for intracranial aneurysm treatment safe and feasible
Alejandro M Spiotta, Department of Neurosurgery, Medical University South Carolina, Charleston, USA, reported his team's experience with the new balloon-stent technique for intracranial aneurysm treatment using the Scepter C balloon catheter and the LVIS, a low-profile visualised intraluminal stent.
Sorin acquires Neurotech
Neurotech designs cost-effective active implantable medical devices used in the treatment of neurological, psychiatric and related disorders.
Age is significantly related to mild cognitive impairment, study says
Memory and executive screening, developed by Zhen Hong, Huashan Hospital, Shanghai Medical College, Shanghai, China, may be a suitable screening method for mild cognitive impairment and mild dementia.
Patients with frontal lobe epilepsy may have effective compensatory mechanism to...
A study lead by Matthias Koepp, UCL Institute of Neurology, London, UK, used fMRI to determine how frontal lobe epilepsy affects the functional brain and memory.
Vitamin B reduces white matter hyperintensities progression in patients with severe...
Daily vitamin B supplementation in patients with severe cerebral small vessel disease (CSVD) significantly reduced white matter hyperintensities (WMH) progression, study shows.
Small study examines ORx calculated by brain tissue oxygenation values from...
Evidence supports that Liscox and Neurovent-PTO probes do not measure ORx equivalently in traumatic brain injury and subarachnoid haemorrhage patients
Asahi Intecc announces US and European launch of Asahi Chikai and...
Asahi Chikai and Asahi Chikai10 were developed as workhorse guidewires to treat intracranial aneurysms, cerebral arteriovenous fistulas and cerebral arteriovenous malformations and provide the torque response, tip flexibility and device deliverability required for these treatments.
Study investigates link between poor physical performance and dementia in 90...
Results suggest strong association of modest declines in physical performance increased risk of dementia.
Phenox receives CE mark approval for p64 Flow Modulation Device
The p64 Flow Modulation Device is an intraluminal flow diverter for treatment of complex intracranial aneurysms and dissections.
Progenitor cells found in the human adrenal medulla may be used...
A research team in Europe has found a way to obtain adrenaline and enkephalin from adult humans and then isolate and force them to become neurons in the lab. This brings researchers one step closer to finding new treatments for neurodegenerative diseases and chronic pain.
Solidarity is power declares World Stroke Organization on World Stroke Day
The World Stroke Organization (WSO) is calling on World Stroke Day (29 October) for stroke communities worldwide to unite and demand accountability from national and regional health authorities in addressing the growing burden of stroke.
Acandis Aperio 1.9F Thrombectomy System receives the CE mark approval
The Acandis Aperio 1.9F Thrombectomy System (Acandis) is a next-generation self-expanding blood clot retrieval system used to treat patients with ischaemic strokes.
Diagnostic accuracy of CEMRA and CTA do not differ significantly in...
A study undertaken in The Netherlands has found that the choice to use contrast enhanced magnetic resonance angiography (CEMRA) or computed tomographic angiography CTA to diagnose intracranial aneurysms in patients presenting with subarachnoid haemorrhage depends on preference or availability however CTA is still preferred.
Serotonin reuptake inhibitors linked to intracerebral and intracranial haemorrhage
Researchers in Canada have found that use of selective serotonin reuptake inhibitors (SSRI) is linked to an increased risk of intracerebral and intracranial haemorrhage, but that the risk is low.
Stroke is becoming more common in young people, new study finds
Brett Kissela, University of Cincinnati College of Medicine in Ohio, USA, and colleagues have found that stroke incidence may be affecting younger adults. The study is published in the 10 October online issue of Neurology.
Steris and Neurologica to collaborate in imaging for neurosurgical procedures
The goal of these joint efforts is to help healthcare providers expand their procedural capabilities, through improved workflow and cost-effective approaches for neurosurgical therapies.
Stryker to acquire Surpass Medical
Surpass Medical develops next-generation flow diversion stent technology to treat brain aneurysms using a unique mesh design and delivery system.
Significant recovery of motor and neurological functions in ischaemic stroke rats...
Neuralstem has reported positive clinical outcomes for Neuralstem's NSI-566 spinal cord-derived neural stem cell line in a rat model of ischaemic stroke. Data has shown significant improvement in both motor and neurological tests in the stem cell-treated rats.
Elekta and Brainlab collaborate to enhance radiosurgery planning power
Elekta and Brainlab announced a collaboration which will enable neurosurgeons and radiation oncologists to use Brainlab's iPlan software together with Elekta's Leksell GammaPlan treatment planning for Gamma Knife radiosurgery.
Infertility treatments may significantly increase multiple sclerosis activity
Jorge Correale, Buenos Aires, Argentina, and colleagues have reported that women with multiple sclerosis who undergo assisted reproduction technology infertility treatment are at risk for increased disease activity.
Use of stent-based thrombectomy may result in a shift towards more...
A small non-randomised study published in Stroke have found that patients with proximal middle cerebral artery occlusion treated with stent-based thrombectomy had more favourable outcomes than patients treated with intravenous tissue plasminogen activator (tPA).
Researchers identify potential new neuroprotective agent for stroke patients
Scientists in Canada have found that a drug called NA-1 results in fewer brain lesions in patients who have undergone surgery to repair brain aneurysms, compared to placebo, and appears to be safe to use.
St Jude Medical’s peripheral nerve stimulation study confirms benefit for chronic...
Data published in Cephalagia shows significant reductions in pain, headache days and migraine-related disability.
MRI Interventions and Brainlab launch European commercialisation of ClearPoint Neuro Intervention...
Stephan Chabardes, Universite Joseph Fourier (UJF) Hospital in Grenoble, France, used the ClearPoint system, for the first time in Europe, to assist with the implantation of deep brain stimulation leads in two patients suffering from Parkinson's disease.
Wellness Center USA announces development of Neurotherapy Devices Group
The Neurotherapy Devices Group will include neuropsychologists, physiologists and medical device engineers who will develop devices and apps intended for use in both home and in-office settings to address brain-based behavioral health disorders without the use of drugs.
SAMMPRIS trial update: only a small subset of patients may be...
A study published ahead-of-print in Stroke has identified risk factors for the increased risk of stroke with percutaneous transluminal angioplasty and stenting that was seen in the SAMMPRIS study.
Vercise Deep Brain Stimulation System gets the CE mark for Parkinson’s...
According to Boston Scientific, developer of the Vercise DBS System, this is the first and only commercially available DBS system to incorporate multiple independent current control, which is designed to selectively stimulate targeted areas in the brain.
UK’s NICE issues final guidance on the use of alteplase to...
NICE has recommended alteplase within the NHS as long as treatment is started as early as possible within 4.5 hours after onset of stroke symptoms, and after intracranial haemorrhage has been excluded by appropriate imaging techniques.
Monarch trigeminal nerve stimulation system for epilepsy, depression to be presented...
The first public presentation of long-term data on the efficacy of Monarch eTNS for the treatment of epilepsy will be shown in a Satellite Symposium sponsored by NeuroSigma.
Eon family of neurostimulators and Genesis neurostimulation system get the CE...
The Eon and Genesis systems (St Jude Medical) deliver peripheral nerve stimulation of the occipital nerves to manage the pain and disability associated with intractable chronic migraine.
FDA clears Sleep Profiler for neuro-cardio assessment of sleep quality
The US Food and Drug Administration (FDA) has granted 510(k) clearance to Advanced Brain Monitoring for its Sleep Profiler, a patient-friendly neuro-cardio diagnostic tool to assess sleep architecture and continuity.
Thrombolysis within six hours after stroke key to improve functional outcome
Results from the IST-3 study, presented at the European Stroke Conference in Lisbon, found that for the type of patients recruited, despite the early hazards, thrombolysis within six hours improved functional outcome in acute ischaemic stroke patients.
FDA approves pivotal study of Smartpatch Peripheral Nerve Stimulation System for...
The Smartpatch PNS System offers a medical device pain relief therapy that has the potential to be an alternative to pain medications, and their accompanying side-effects.
Registry shows positive outcomes for the Codman Enterprise device
New registry data shows 81% of patients had 90% or higher aneurysm occlusion after 21 months and only 8.3% required retreatment after stent-assisted coil embolization with the Codman Enterprise Vascular Reconstruction Device (VRD) and Delivery System (Codman Neurovascular).
FDA approves new multiple sclerosis treatment Aubagio
Aubagio (teriflunomide, Bridgewater) is a once-a-day tablet for the treatment of adults with relapsing forms of multiple sclerosis.
Study says new self-expanding clot retrieval system seems efficient and safe
A small, experimental study, published in the American Journal of Neuroradiology (AJNR) in August, suggests that the Aperio System (Acandis Medical) seems to be an "efficient and safe" addition to the existing repertoire of clot-removal devices.
Penumbra Coil 400 approved in Japan for treatment of intracranial aneurysms
According to Penumbra, this coil is capable of tighter packing, and thus greater occlusion density, which can improve long-term outcomes.
FDA approves higher dose of Exelon Patch for patients with mild...
The approval is based on the drug's efficacy and safety demonstrated in the OPTIMA long-term study comparing 13.3mg/24 h to 9.5mg/24 h in declining mild to moderate Alzheimer's patients.
Elimination of brain enzyme may be a new target for Alzheimer’s...
Scientists in the USA have found that eliminating an enzyme called jnk3 from mice with symptoms of Alzheimer's disease leads to a 90% reduction in the compounds responsible for formation of the plaques linked to Alzheimer's disease.
Stem cell graft restores memory and mood
Grafting stem cells from one part of the brain into another can restore important cognitive functions such as memory and mood after brain injury and also trigger the growth of new neurons, a US research team has found.
InSightec receives FDA approval to begin phase I Parkinson’s trial of...
Thirty patients who suffer from medication-resistant tremor of Parkinson's disease will be treated in a randomised control trial and followed-up for one year.
UK’s NICE issues draft guidance on the use of altelplase to...
NICE has recommended alteplase within the NHS as long as treatment is started as early as possible within 4.5 hours after onset of stroke symptoms, and after intracranial haemorrhage has been excluded by appropriate imaging techniques.
First-in-man use of the Acandis Acclino 1.9F Stent System
With sizes compatible for vessel diameters of 2 - 4 mm, the laser cut nitinol stent is suitable for all of the cerebral main vessels and those even more distal and rather difficult to reach, where a low profile delivery system is essential.
Codman Neurovascular launches two new microcoil systems, new distal access catheter...
Codman Neurovascular has announced the launch of the Deltamaxx Microcoil System, and Orbit Galaxy G2 Microcoils, which for the first time puts Orbit Galaxy coils on the Enpower Detachment System, developed by Micrus Endovascular.
New mobile software enables physicians to consult on stroke cases anytime,...
First-of-its-kind mobile phone-based software technology may allow physicians to manage and consult on stroke cases in real time from anywhere in the world, a study presented at the Society of NeuroInterventional Surgery (SNIS) 9th Annual Meeting in San Diego, USA, showed.
Do pre-specified ASPECTS scores predict good outcome for acute ischaemic stroke...
Interim results from the START trial, presented at Society of NeuroInterventional Surgery (SNIS, San Diego, USA) 9th Annual Meeting, show advancement in predicting which patients suffering from ischaemic stroke will benefit from endovascular therapy.
FDA approves Medstream Programmable Infusion System for treatment of spasticity
The Medstream system, already approved in Europe, delivers a continuous and highly accurate dose of the anti-spasm drug baclofen directly to the spinal canal to relieve the symptoms of severe spasticity.
ADvance study starts patient enrolment to assess deep brain stimulation for...
ADvance is a randomised double-blind controlled trial initially involving 20 people, in Canada and USA, aged 55-80 with mild Alzheimer's disease.
First patient enrolled in stem cell therapy study for stroke
A new US study looking at the safety and effectiveness of new medication developed from adult stem cells for the treatment of ischaemic stroke has opened at University Hospitals (UH) Case Medical Center, Cleveland, USA.
Hydrogel endovascular aneurysm treatment (HEAT) clinical trial starts patient enrolment
This prospective, randomised, international multicentre trial is designed to compare clinical outcomes in patients with intracranial aneurysms treated with the new generation of the MicroVention hydrogel coils to those treated with bare platinum coils.
Brain aneurysms linked to early onset menopause, study finds
A correlation between brain aneurysms, bulges or balloons in vessel walls, and early onset menopause has been identified, according to recent study results presented at the Society of NeuroInterventional Surgery (SNIS) 9th Annual Meeting (San Diego, USA, 23-27 July).
FDA approves BrainsGate stroke-treatment study
BrainsGate's stroke-treatment is designed to treat ischaemic strokes with 24 hours of an event. Current pharmaceutical treatments, which include tPA proteins, are permitted only within 3-4.5 hours of a stroke.
Advances may make time window less important in stroke treatment
A multicentre study evaluated the use of computed tomography (CT) perfusion imaging, regardless of time from symptom onset, in selecting patients for endovascular treatment with devices or clot-busting drugs used directly at the problem site in the brain.
Barricade Coil System gets CE mark
The Barricade Coil System (Blockade Medical) is designed to occlude blood flow in vascular abnormalities of the neurovascular and peripheral vessels.
Including stroke severity in risk models associated with improved prediction of...
Adding stroke severity to a hospital 30-day mortality model based on claims data for Medicare beneficiaries with acute ischaemic stroke was associated with improvement in predicting the risk of death at 30 days and changes in performance ranking regarding mortality for a considerable proportion of hospitals, according to a study published by Gregg C Fonarow, University of California, Los Angeles, USA, and colleagues.
Preliminary results with the Web aneurysm device for bifurcation aneurysms
Early evidence with the Web device (Sequent Medical) suggests that this represents a feasible and promising treatment for wide-neck bifurcation aneurysms, and that further study and clinical experience is warranted.
Lazarus Effect Funnel Guide Catheter receives CE mark
This catheter is designed to enhance the introduction and removal of interventional devices, including those used to treat acute ischaemic stroke.
Interferon beta not linked with less progression of disability in relapsing-remitting...
Afsaneh Shirani, University of British Columbia, Vancouver, Canada, and colleagues have found that treatment with interferon beta was not associated with less progression of disability among patients with relapsing-remitting multiple sclerosis.
NeuroLogica announces integration with Medtronic Surgical Navigation Solutions
NeuroLogica and Medtronic Surgical Technologies have announced the integration of NeuroLogica's portable BodyTom 32-slice CT scanner with Medtronic's StealthStation S7 surgical navigation system.
Stress management training may help reduce disease activity in multiple sclerosis...
A new study published by David C Mohr, Northwestern University Feinberg School of Medicine in Chicago, USA, and colleagues shows that taking part in a stress management programme may help people with multiple sclerosis (MS) prevent new disease activity.
Neurostimulation devices market expected to grow
A new report by GBI Research predicts that the neurostimulation devices market will reach US$5,305.5m by 2018, following a climb at a compound annual growth rate (CAGR) of 14.1% from 2011.
Natural course of unruptured cerebral aneurysms varies according to the size,...
A study from Japan, published in late June in the New England Journal of Medicine, provides information on the natural history of unruptured cerebral aneurysms from over 6,600 aneurysms.
Hormonal treatment associated with better test performance after stroke
Stroke patients who received hormonal treatment, combined with rehabilitation, performed better on functioning and reasoning tests than patients who received rehabilitative therapy alone, a new clinical study from Italy shows.
NeuroSigma receives global quality control certification for its external Trigeminal Nerve...
The external Trigeminal Nerve Stimulation (eTNS) system called theMonarch is designed for treatment of epilepsy and depression. This certification is an important first step in a two-step process towards CE mark approval in the European Union.
Nexstim gets FDA clearance for NexSpeech
NexSpeech, designed to be used with the Navigated Brain Stimulation System (Nexstim), enables noninvasive speech mapping prior to neurosurgery.
Mount Sinai brings benefit of robotic surgery to sleep apnea sufferers
Through the robotic procedure, a laser removes the extra tissue in the throat that contributes to the airway obstruction in sleep apnea patients. Patients typically return home the next day, and are back to work in 10 days, sleeping and breathing normally.
Deep brain stimulation helps Parkinson’s patients over long term
A multicentre, randomised, controlled trial found that deep brain stimulation produced a beneficial effect on the motor function of Parkinson's disease patients over the course of three years.
Results of levodopa-carbidopa intestinal gel study for Parkinson’s disease presented
A second interim analysis of a long-term safety and tolerability trial, as well as secondary endpoint analyses from the phase III pivotal trial evaluating the use of Abbott's levodopa gel for Parkinson's disease patients were presented at the International Congress of Parkinson's Disease and Movement Disorders (17-21 June, Dublin, Ireland).
American Academy of Neurology issues new guideline for treating infantile spasms
The guideline which is based on a review of all available evidence on treatment for infantile spasms and is an update of a guideline published in 2004, found that adrenocorticotropic hormone therapy may be effective for treatment of infantile spasms.
GE Healthcare, Australian government further research into Alzheimer’s
GE Healthcare will provide its investigational imaging agent [18F flutemetamol] to the Australian governmental agency (CSIRO) for use in the Australian Imaging, Biomarker & Lifestyle Flagship Study of Ageing (AIBL).
Penumbra launches the Max System Reperfusion Catheters for acute ischaemic stroke...
The Max system offers a fast, simplified cerebral thrombectomy procedure with single-operator, over-the-wire delivery, without the need for a large proximal balloon guide.
Smelling test may help to detect Parkinson’s at an early stage
Smelling tests conducted in public places have yielded impressive results, neurologists from Cologne, Germany, reported at the meeting of the European Neurological Society (Prague, Czech Republic, 9-12 June 2012).
Strokes among young people often overlooked, international study finds
Strokes are not just a medical condition of the advanced age - younger people can also be affected. But in this group, many strokes remain "silent" or symptoms are being misinterpreted. These findings from a large-scale European study were presented at the 22nd Meeting of the European Neurological Society.
Mazor Robotics announces first brain cases using its robotic platform
Mazor expands clinically validated technology from spine into guided brain operations
Stem cell therapy is promising, but neurologists warn about unscrupulous providers
According to experts at the 22nd Meeting of the European Neurological Society, current research findings give reason to hope that different types of stem cells could open up new prospects in therapy for severe neurological diseases such as stroke, Parkinson's or multiple sclerosis. But given the many unresolved issues, neurologists warn about dangerous promises of cures from unscrupulous providers.
New methods for better coma diagnosis
Calling patients in coma-like states by name is a simple and effective way of assessing what consciousness functions they still possess. Their own name triggers reactions in visible behaviour and in measurable brain activity not otherwise unleashed by less personal stimuli.
Orthofix International announces integration with Brainlab
Orthofix will integrate its Phoenix MIS and Firebird Pedicle Screw Systems into the latest generation of Brainlab spinal navigation systems.
Taking the stroke unit to the patient reduces time from alarm...
A randomised controlled trial published in The Lancet Neurology in May has shown that for patients with suspected stroke, treatment in a mobile stroke unit substantially reduced median time from call for help to therapy decision.
Need for emergency neurosurgical procedures after intracranial endovascular treatment is low
Rakesh Khatri and colleagues at the Zeenat Qureshi Stroke Research Center, University of Minnesota, USA, have recently published a study showing that despite the fact that provision for emergency neurosurgical procedures is a mandatory component of centres that perform neuroendovascular procedures, the need for them is quite low.
GlobalData releases report on Japanese neurology devices market outlook to 2018
The report contains information on interventional neurology devices, neurological diagnostic equipment and neurostimulation devices.
Trevo Retriever performs better than Merci Retriever, TREVO 2 study shows
Results from the TREVO 2 clinical trial were presented by Raul Nogueira, Atlanta, USA, at the European Stroke Conference (Lisbon, Portugal, 22-25 May 2012).
Minimus Spine receives notice of allowance for pre-sterilised vial to create...
This pre-sterilised vial that interfaces with a console to create ozone gas can be used as part of a non-surgical treatment for disc herniations.
Is delayed haemorrhage a complication following treatment with a flow diverter?
A recently published study reports that delayed ipsilateral parenchymal haemorrhage may occur after the treatment of anterior circulation aneurysms with flow diverters. The study was published in the American Journal of Neuroradiology.
Nerve transfers help to restore some hand function in quadriplegic patient
Susan E Mackinnon (Washington University School of Medicine, St Louis, USA) and colleagues have reported the first case of restoration of some hand function (finger and thumb flexion) in a quadriplegic patient utilising nerve transfers after cervical spinal cord injury.
Freedom Cervical Disc gets CE mark
AxioMed Spine has received CE mark approval for its Freedom Cervical Disc, an elastomeric total spinal disc replacement device.
NICE’s draft guidance recommends Botox for adults with chronic migraine who...
The draft guidance recommends that Botox injections should be stopped if the person's headaches have not improved enough after two treatment cycles, or if the person's "headache days" have reduced to fewer than 15 days a month over three consecutive months.
Intestinal gel reduces ‘off’ time in advanced Parkinson’s disease
The intestinal gel contains levodopa and carbidopa and is infused through a portable pump connected to a tube implanted in the intestine, similar to a feeding tube.
Eating low-fat dairy foods may reduce risk of stroke
A Swedish study of 74,961 adults with 10 years of follow-up has shown that those who ate low-fat dairy foods had a 12% lower risk of stroke and a 13% lower risk of ischaemic stroke than those who ate high-fat dairy foods.
Use of interferon beta-1a after first sign of possible multiple sclerosis...
Phase three results of the REFLEXION clinical trial have shown that people who received injections of the interferon beta-1a drug soon after their first signs of possible multiple sclerosis were less likely to progress to clinically definite multiple sclerosis than people who switched to interferon beta-1a from placebo.
Analysis of the CLARITY series shows no difference between Guglielmi Detachable...
Laurent Pierot and colleagues, on behalf of the CLARITY investigators, reported that midterm anatomic results and evolution of aneurysm occlusion were not different in patients with ruptured aneurysms who were treated with Guglielmi Detachable Coils (GDC) when compared to those treated with Matrix coils.
American Academy of Neurology releases new guidelines to help prevent migraine
Research shows that many treatments can help prevent migraine in certain people, yet few people with migraine who are candidates for these preventive treatments actually use them, according to new guidelines issued by the American Academy of Neurology.
NeuroLogica, Stryker announce partnership
NeuroLogica and Stryker Navigation, a business unit of Stryker, have announced the development of an integrated surgical navigation unit for NeuroLogica's portable BodyTom 32-slice CT scanners.
1 Mission 1 Million initiative works to improve stroke prevention in...
1 Mission 1 Million - Getting to the Heart of Stroke announced plans to launch a new phase of the initiative dedicated to raise awareness of atrial fibrillation and its link to stroke.
Aging accelerates brain abnormalities in childhood onset epilepsy patients
New research confirms that childhood onset temporal lobe epilepsy has a significant impact on brain aging.
Actilyse authorised in the UK for treatment of ischaemic stroke patients...
The extended treatment window from 3 to 4.5 hours will allow a greater number of eligible patients to receive thrombolysis.
Unruptured aneurysms affect quality of life
Patients with unruptured brain aneurysms have significant reductions in several areas of quality of life-especially physical functioning, according to a study published in the April issue of Neurosurgery.
BodyTom CT scanner gets CE mark approval
NeuroLogica Corporation announced that it has received CE mark approval for its BodyTom, a portable, full body, multi-slice CT scanner.
Medtronic gets Health Canada licence for deep brain stimulation therapy in...
The licence was based on data collected from the SANTE trial, a prospective, randomised, double-blind pivotal study which evaluated the use of DBS therapy in patients with medically refractory epilepsy with partial-onset seizures.
Intracranial aneurysms treated with the Silk flow diverter have high occlusion...
A study presented at the European Congress of Radiology (ECR) in Vienna, Austria, showed that flow diverter treatment of complex aneurysms is effective with a high aneurysm occlusion rate at one year follow-up, despite a complication rate that was "not low".
NeuroSigma appoints UCLA neurologist as vice-president of Neurology
Christopher M DeGiorgio will lead NeuroSigma's development of Trigeminal Nerve Stimulation (TNS) for the treatment of neurological indications, such as drug-resistant epilepsy.
Inceptus Medical gets licence to use Sequent Medical’s MicroBraid technology
Sequent Medical's WEB Aneurysm Embolization System utilises MicroBraid technology in the treatment of cerebral aneurysms.
Siemens Healthcare to showcase Hybrid molecular MR imaging solution and PET...
The Biograph mMR is a hybrid molecular MR and the only system in the medical technology marketplace that combines the functionality of MR and PET in a single integrated solution.
SanBio announces enrolment of first cohort of patients in clinical trial...
The therapy called SB623 is derived from adult bone marrow and has shown safety and efficacy in rodent models of chronic stroke.
SPS3 results do not support use of combination clopidogrel plus aspirin...
Interim results from the SPS3 (Secondary prevention of small subcortical strokes) study, presented by Oscar Benavente, Vancouver, Canada, revealed that combination therapy using clopidogrel plus aspirin is not effective in preventing secondary strokes over the long term, in patients with small subcortical strokes.
Aggressive medical therapy better than stents, even for patients who failed...
A subanalysis of the SAMMPRIS trial found that there were lower outcome events with aggressive medical management than with stenting, even in patients who had failed antithrombotic therapy, who are perceived as being at high-risk.
Zynex acquires NeuroDyne Medical
NeuroDyne Medical is an 18 year old manufacturer of advanced medical devices for non-invasive measurement of sEMG and autonomic nervous systems.
First patient enrolled into ‘MORE’ European registry for deep brain stimulation...
The primary objective of the registry is to examine the impact of Medtronic's deep brain stimulation therapy on seizure frequency. The study will also look at seizure type and severity, safety and impact of the therapy on a patient's quality of life.
Solitaire FR revascularisation device receives FDA clearance
The Solitaire FR revascularisation device (ev3 / Covidien) is intended to restore blood flow to the brain in patients suffering acute ischaemic stroke by mechanically removing blood clots from blocked vessels. The clearance was supported with results from the SWIFT trial.
The Brain Aneurysm Foundation announces new Medical Advisory Board members
The new members are Bernard R Bendok, Northwestern University Feinberg School of Medicine, Paul J Camarata, University of Kansas, and Aaron A Cohen-Fadol, Goodman Campbell Brain and Spine.
New drug may block the effects of traumatic brain injury
Preliminary data from a study in rats found that clazosentan decreased the effects of the traumatic brain injury on blood flow to the hippocampus by 25% at four hours and 23% at 48 hours after traumatic brain injury.
Solitaire FR device approved for interventional management of stroke trial (IMS...
The IMS III trial will compare a combined intravenous and intra-arterial treatment approach to restoring blood flow to the brain to the current standard FDA-approved treatment approach of giving IV rTPA alone.
Men with strokes call for help quicker than women
Married men called within 28 minutes of symptoms compared to married women, who called an average of 67 minutes after their first stroke symptoms, according to a Mayo Clinic study presented at the 2012 International Stroke Conference, New Orleans, USA.
Novel drug shows significant promise in treating severe stroke patients
Preliminary results of a phase 2 clinical trial using Remedy Pharmaceuticals' new drug, RP-1127, presented at the 2012 International Stroke Conference, have shown positive results in the treatment of patients with severe stroke, prompt to develop significant brain swelling.
SWIFT trial results presented at International Stroke Conference
Results from the SWIFT (Solitaire with the intention for thrombectomy) study show that the Solitaire FR revascularisation device (ev3 / Covidien) opened blocked vessels without causing symptomatic bleeding in or around the brain in 61% of patients, compared to 24% of cases performed with the FDA-approved Merci retrieval system (Concentric Medical / Stryker).
Clot-busting drugs appear safe for treating “wake-up” stroke patients
A study presented at the American Stroke Association's International Stroke Conference 2012 shows that clot-busting drugs may be safe for patients who wake up experiencing stroke symptoms.
TREVO results presented at the International Stroke Conference
Results from the prospective, multicentre clinical study show that 55% of patients who suffered a severe acute ischaemic stroke were restored to a good functional outcome.
Anaemia may triple the risk of dying after a stroke
Being anaemic could more than triple the risk of dying within a year after having a stroke, according to research presented at the American Stroke Association's International Stroke Conference 2012.
Sleep apnea linked to silent strokes
People with severe sleep apnea may have an increased risk of silent strokes and small lesions in the brain, according to a small study presented at the American Stroke Association's International Stroke Conference 2012.
Severe, rapid memory loss linked to future, fatal strokes
Researchers found that people who died after stroke had more severe memory loss in the years before stroke compared to people who survived stroke or people who did not have a stroke.
Clazosentan reduces “second stroke” after aneurysm rupture
A randomised trial (CONSCIOUS-3) shows that treatment with clazosentan, an investigational endothelin receptor antagonist, reduces de risk of vasospasm after aneurysm rupture.
Infections in childhood linked to high risk of ischaemic stroke
Researchers in USA have found that acute infections in children are more important in triggering ischaemic stroke than chronic infections.
Solitaire FR approved for interventional management of stroke trial
The Solitaire FR revascularisation device (Covidien) has been approved for investigational use in the thrombectomy arm of the IMS III (Interventional management of stroke) trial.
Penumbra launches Separator 3D at the ABC-WIN seminar
The Separator 3D is a next-generation clot engagement device that works in conjunction with aspiration at the site of the lesion causing an acute ischaemic stroke to remove the clot and restore blood flow to affected brain tissue.
ALS phase I/II clinical trial shows positive results for NurOwn stem...
The ALS phase I/II human clinical trial is being performed at Hadassah Medical Center in Israel in collaboration with BrainStorm and is utilising NurOwn technology (BrainStorm) for growing and modifying autologous adult human stem cells to treat ALS.
Enrolment in first MRgFUS essential tremor feasibility trial completed
Fifteen patients underwent a non-invasive magnetic resonance guided focused ultrasound treatment to evaluate safety and initial effectiveness of the Exablate Neuro (InSightec).
Randomised controlled study of deep brain stimulation confirms benefit of constant...
Results from the first St Jude Medical controlled study on deep brain stimulation for Parkinson's disease show statistically significant increases in symptom control and improved quality of life. Data were published in the January issue of Lancet Neurology.
Clarimedix secures investment for proof-of-concept study in cerebral vasospasm
Clarimedix announced that Johnson & Johnson Corporate Office of Science and Technology (COSAT) has secured an investment for a human proof-of-concept study to determine the viability of a powerful nitric oxide donor alternative for the treatment of cerebral vasospasm, Raynaud's phenomenon, and Alzheimer's disease.
Mobiles and social media set to transform stroke communication
New research from Japan suggests that hand-held communication devices could have an impact on stroke diagnosis and management.
MindFrame awarded new patent for breakthrough Capture LP thrombectomy technology
MindFrame announced the issuance of US Patent No. 8,088,140, bringing the total number of patents awarded for its Capture LP thrombectomy technology for ischaemic stroke therapy to three.
First 20 patients enrolled in the InterSEPT clinical trial to study...
InterSEPT data will be used to apply for CE marking for the SeptRx Intrapocket Occluder (IPO)-a platform for the percutaneous transcatheter closure of patent foramen ovale.
MindFrame gets CE mark for new Capture LP devices for treatment...
The low profile devices have been expanded to include a 4-mm diameter and shorter length sizes.
Ischaemic stroke registry to start
The goal of MindFrame's sponsored registry is to support clinicians in advancing knowledge around the use of mechanical thrombectomy technologies for the treatment of patients suffering from ischaemic stroke.
First procedure with Brainlab’s Curve image guided surgery system
University hospital Klinikum rechts der Isar, Munich, Germany, announced that this is the first hospital operating with Brainlab's Curve image guided surgery system. This new technology provides surgeons with better guidance and control during surgery enabling faster, more precise and safer interventions.
Scientists identify a novel therapy with potential for treating Parkinson’s disease
A collaboration between virologists and neuroscientists at Cambridge University, UK, has demonstrated how viruses that cross the blood/brain barrier could be exploited to slow down, or even halt, the progress of Parkinson's and other neurodegenerative diseases.
Devon Medical receives Chinese clearance for ViewSite brain access system
The ViewSite is a brain access and retraction device that allows higher precision for surgeons in reaching target areas in the brain, such as tumors and haemorrhages, in a minimally invasive manner.
BrainScope awarded US$7.5 million US Department of Defense Research contract for...
The Ahead M-100 is a handheld, non-invasive, non-radiation emitting medical device in development that could provide a rapid and objective assessment and categorisation of brain injury, based on patterns identified in brain electrical activity.
Functional brain pathways disrupted in children with attention deficit/hyperactivity disorder, study...
Using functional magnetic resonance imaging (fMRI), researchers have identified abnormalities in the brains of children with attention deficit/hyperactivity disorder (ADHD) that may serve as a biomarker for the disorder.
Encouraging early clinical results with Woven Endobridge cerebral aneurysm device
A study published in the journal Neuroradiology suggests that the intrasaccular deployment of self-expanding, compliant, cylindrical, high-density, braided metallic mesh constructs could represent a reasonable approach as an endovascular treatment of cerebral aneurysms.
86.5% of patients experience chronic pain relief with neurostimulation system, study...
The AdaptiveStim with RestoreSensor neurostimulation system from Medtronic provides clinical benefits for many patients, including effective pain relief and convenience, compared to systems without position-adaptive stimulation technology.
GE Healthcare introduces Innova IGS 630 biplane interventional imaging system
The system features Innova CT HDii 3D technology and a comprehensive set of advanced applications for interventional neuroradiology.
Spinal Modulation Neurostimulator system receives CE mark for the management of...
This system utilises low-level electrical signals to modulate neural structures of primary sensory neurons located within the dorsal root ganglion.
Eyebrain Tracker to be used in clinical trial for Parkinson’s therapy
The trial sponsored by Bordeaux University Hospital will explore the dyskinesia induced by treating patients suffering from idiopathic Parkinson's disease with levodopa.
CareFusion launches wireless diagnostic and monitoring neurological device
The Nicolet EEG Wireless Amplifier aids clinicians in their diagnosis and treatment of epilepsy, brain trauma, stroke, brain tumors, sleep dysfunction and clinical research.
Haemoglobin and blood pressure levels, male gender linked to “silent” strokes...
Researchers from USA have found that lower concentrations of haemoglobin, higher baseline systolic blood pressure, and male gender are associated with a significantly increased risk of silent stroke in children with sickle cell anaemia.
Violent video games alter brain function in young men
The controversy over whether or not violent video games are potentially harmful to users has raged for many years, but there has been little scientific evidence demonstrating that the games have a prolonged negative neurological effect.
“Heading” a soccer ball could lead to brain injury
Using diffusion tensor imaging (DTI) to study the effects of soccer "heading", researchers have found that players who head the ball with high frequency have brain abnormalities similar to those found in traumatic brain injury (TBI) patients.
Is brain bypass becoming a lost art?
A microsurgical procedure that has lost some ground to advances in endovascular therapy still plays a critical role in the management of selected neurovascular disorders, according to Nicholas C Bambakidis, neurosurgeon, University Hospitals Case Medical Center, Cleveland, USA.
Greater number of vertebroplasty patients discharged to rehabilitation centres than home
In a retrospective review, US researchers found that 90 in-patients underwent vertebroplasty, 73 of whom lived independently at home before hospitalisation. Of these 73 patients, 42% returned home after discharge, 12% returned home with healthcare assistance, and 44% were sent to skilled nursing facilities.
Prolonged imaging unnecessary for most adequately coiled intracranial aneurysms
Researchers from The Netherlands found that for the vast majority of adequately occluded intracranial aneurysms six months after coiling, prolonged imaging follow-up within the first five to 10 years after coiling does not seem beneficial for detecting re-opened aneurysms that need re-treatment.
SPR Therapeutics implants world’s first SPR system for chronic pain
The system has been used as part of a multicentre clinical study for the treatment of post-stroke shoulder pain.
First Illinois patient successfully treated in TREVO 2 study
The TREVO 2 study is designed to assess the ability of the Trevo retrieval system (Concentric Medical) to remove the blood clots that cause strokes and to restore blood flow to the brain.
First patient from a next dose cohort successfully treated in stem...
ReNeuron reported that the first patient of three to be treated with a higher dose cohort of ReN001 has been successfully discharged from hospital with no acute safety issues arising.
Crossing legs after severe stroke may be a good sign of...
People who were able to cross their legs within the first 15 days after a severe stroke were more likely to have better independence in daily life, fewer neurologic problems and lower death rates, according to a study published in Neurology.
“Retrospective data shows safety and efficacy of Solitaire FR as a...
Data presented by Jan Gralla, Bern, Switzerland, at CIRSE 2011 in Munich, Germany, showed that Solitaire FR from ev3/Covidien had a good recanalisation rate and good patient outcome at 90 days, as measured by the modified Rankin Scale.
Multicentre, retrospective data shows safety and efficacy of Solitaire FR as...
Data presented by Jan Gralla, Bern, Switzerland, at CIRSE 2011 in Munich, Germany, showed that Solitaire FR from ev3/Covidien had a good recanalisation rate and good patient outcome at 90 days, as measured by the modified Rankin Scale.
Neurosurgical and neurovascular device market to grow at six per cent...
The growth will be driven largely by an aging population in the United States that will suffer an increasing incidence of conditions such as stroke, cerebral aneurysms and intracranial atherosclerosis disease.
Unselected series examines complications after flow diverter usage
Istvan Szikora, Hungary, spoke on behalf of the ESMINT Flow Diverter Bleeding Project contributors to tell ESMINT delegates that a small, real world, unselected series analysis has shown that the characteristics determining delayed rupture of intracranial aneurysms when treated by flow diverters are aneurysm size and symptomatic clinical presentation.
No strict time window or age limit to treat acute ischaemic...
Iris Q Grunwald, Oxford, UK, told delegates at the CIRSE annual meeting that mechanical recanalisation devices allow an extension of the conventional therapeutic time window and can be used without adjuvant thrombolytic therapy thus potentially reducing the intracranial bleeding risk in the treatment of acute ischaemic stroke.
St. Jude gets Japanese approval for neurostimulation lead to manage chronic...
The Penta surgical lead addresses complex pain patterns like low back pain, arm and leg pain.
EFIC launches European week against pain
The campaign is dedicated to raising awareness of chronic back pain, and includes the design of a new and appropriate classification of this condition, the promotion of better research, and the development of more effective treatments.
Bioness receives FDA clearance and CE mark for the Ness H200...
The NESS H200 wireless is an advanced hand rehabilitation system designed to use mild functional electrical stimulation to improve hand function and promote motor recovery in patients who have lost function of their upper extremity following injury to the central nervous system.
Focused ultrasound shows promise as a noninvasive deep brain treatment for...
Results from the study's first 10 patients showed a 78% improvement in contralateral tremor scores in the hand, as assessed with the Clinical Rating Scale for Tremor (CRST). Patients' functional activities scores improved by 92%, as measured in the 'Disability' subsection of the CRST.
Clinical registry shows positive results for Avance Nerve Graft for mixed...
Study results demonstrate that Avance Nerve Graft (AxoGen) is a safe method for reconstructing nerve discontinuities with 87.3% of the injuries achieving meaningful recovery.
Onyx vs. Glue: End the battle!
In a debate on Onyx vs. Glue, Laurent Spelle, Paris, France told delegates at the ESMINT congress in Nice, France, that it was time to lay the battle of Onyx vs. glue to rest. He said "We must quit focussing on Onyx vs. glue-each material has its own proper indications, and we should focus our efforts to better understanding how arteriovenous malformations behave."
Can CT perfusion guide patient selection for endovascular treatment of acute...
CT perfusion-guided patient selection for endovascular treatment of acute ischaemic stroke is safe and effective, report Aquilla Turk, Medical University of South Carolina, Charleston, USA and colleagues in a study published in September in the Journal of NeuroInterventional Surgery.
Zynex gets FDA 510(k) clearance for NexWave electrotherapy device
The NexWave device delivers three modalities of stimulation; traditional transcutaneous electrical nerve stimulation (TENS), interferential and neuromuscular electrical stimulation for pain and muscle rehabilitation therapy.
Concentric Medical completes enrolment for mechanical thrombectomy in acute ischaemic stroke...
"The TREVO study represents an important step forward in the evidence supporting the treatment of patients with moderate and severe strokes with mechanical thrombectomy devices, such as the Trevo system (Concentric Medical/Stryker)," according to Nils Wahlgren, co-principal investigator of the study.
30 patients with chronic neurological disorders have been treated with ExAblate...
These patients are participating in clinical studies conducted in Switzerland and the USA to evaluate the safety and initial efficacy of Exablate Neuro (InSightec) to treat essential tremor, neuropathic pain and Parkinson's disease.
First patients enrolled in clinical study of portable brain-sensing system for...
"Our Nautilus NeuroWave system (Jan Medical) can rapidly provide critical information on a patient presenting with stroke symptoms, and it can also be used as a continuous monitor of changes to the cerebral vasculature," Paul Lovoi, CEO of Jan Medical.
US National Institutes of Health gives notice of award to fund...
This award will provide total funding of approximately US$750,000 to NeuroSigma, worldwide licensee of the eTNS therapy (external trigeminal nerve stimulation) which has been developed by the University of California.
New cardiopulmonary resuscitation device improve long-term brain function following cardiac arrest
Results from the ResQTrial demonstrated that the ResQCPR system (Advanced Circulatory Systems) when used together with therapeutic hypothermia had a six-fold increase in the percentage of patients who improved from poor neurologic function at hospital discharge to good neurologic function at 90 days, when compared to standard CPR with hypothermia.
Genesis neurostimulation system gets CE mark for the treatment of chronic...
The CE mark approval of Genesis (St Jude Medical) was supported by the results of a randomised, double-blind, controlled, chronic migraine study of 157 patients who suffered from headache, on average, 26 days per month.
Calcified plaque in arteries may cause stroke or dementia
New research shows that calcium build-up in arteries outside the brain may be associated with brain changes linked to future risk of dementia and stroke.
Brain Attack Coalition updates recommendations for primary stroke centres in the...
The new recommendations recognise substantial changes in the diagnosis and treatment of stroke patients over the past decade particularly advances in brain and imaging techniques, the development of stroke teams and stroke units, and evidence supporting the importance of early initiation of rehabilitation therapy.
MAPS results positive for coiling aneurysms
In the Matrix and platinum science (MAPS) trial, 96% of patients with unruptured aneurysms and 90% of patients with acutely ruptured aneurysms were alive and free of disability out to 15 months after treatment.
Stryker to acquire Concentric Medical for US$135 million
The acquisition of Concentric provides Stryker immediate access into the acute ischaemic stroke market segment in the USA, Europe and Japan.
Alex M DiNello is the new president and CEO of Relievant...
DiNello has 18 years experience in spinal products' development and comercialisation; he will lead Relievant Medsystems just after the FDA's IDE approval for the assessment of its Intracept system, a radiofrequency ablation therapy for the treatment of vertebrogenic back pain.
Minimus Spine receives notice of allowance for its syringe designed to...
"The syringe will give the physician and patient confidence that the injection is done in a sterile manner," said David Hooper, Minimus Spine founder and CEO.
Zynex to acquire NeuroDyne Medical assets
NeuroDyne Medical produces devices for evaluation and treatment of neurological and neuromuscular disorders.
GE Healthcare imaging technology aids in Parkinson’s disease diagnosis
DaTscan (GE Healthcare) is the only FDA-approved imaging agent for assessment of movement disorders. This technology allows doctors to differentiate Parkinson's from other movement disorders.
Improved diagnostics could reduce risky surgery for asymptomatic carotid stenosis
New Canadian research has shown that using 3D ultrasound to identify ulcers in the carotid arteries is an effective way to pinpoint the small number of high-risk patients with asymptomatic carotid stenosis who would benefit from surgery to prevent stroke.
Study explores impact of more uniform distribution of endovascular coils
A more uniform distribution of endovascular coils may help in the treatment of cerebral aneurysms. US study investigators Bernard R Bendok and Matthew Gounis spoke to NeuroNews on their findings.
First-in-human procedure performed with the MindFrame LP device
Recently CE marked, the MindFrame LP devices are designed to treat patients suffering from ischaemic stroke through microcatheter-based flow restoration and clot extraction.
Cognitive behavioral therapy may help people with unexplained symptoms of pain,...
A UK study showed that after three months, people who received cognitive behavioral therapy as an addition to their usual care were approximately twice as likely to report improvements in their overall health as those who received just the standard usual care.
Cholesterol-lowering drugs may help prevent recurrent strokes in young people
Researchers from Finland found that patients who were treated with a statin at any time after their first stroke were 77% less likely to experience another stroke or vascular problems compared to those not treated with a statin at all.
Oxygen Biotherapeutics partner with Aurum Biosciences to explore the use of...
The intent of this research is to better delineate which tissue has been damaged from a stroke, and to determine the effectiveness of the treatment being provided to the model.
High blood pressure, diabetes, smoking and obesity in middle age may...
"These factors appeared to cause the brain to lose volume, to develop lesions secondary to presumed vascular injury, and also appeared to affect its ability to plan and make decisions as quickly as 10 years later," said study author Charles DeCarli, University of California, USA.
AccuTrack 3D image guidance tool gets Health Canada approval
AccuTrack (IMRIS) aids surgeons in critical decision making by utilising an ergonomic lightweight probe to quickly and accurately localise target anatomy before; and at any time during a neurosurgical procedure.
New study reveals negative financial impact of some neurovascular treatments on...
A study shows that treatment costs for some neurovascular conditions remain higher than reimbursement in the form of Medicare payments in the USA.
MindFrame LP devices receive CE mark
The MindFrame Capture LP and MindFrame Flow LP devices are designed to treat patients suffering from ischaemic stroke through microcatheter-based flow restoration and clot extraction, allowing for rapid perfusion and thrombus retrieval.
PUFS clinical study results show device safety and effectiveness
Data presented at the Society of NeuroInterventional Surgery 8th Annual Meeting showed that the Pipeline embolization device (Covidien) met the trial primary endpoints for both effectiveness and safety.
THERAPY trial will assess benefits of catheter-based intervention in the treatment...
This randomised controlled trial could significantly impact the delivery of stroke treatment services in hospitals across the USA, investigators say.
Radiosurgery treatment for brain tumours available for UK patients
Novalis Tx (Varian Medical Systems and Brainlab), a non-invasive system to treat brain tumours with radiosurgery has gone into service at The Christie at Salford Royal, a brand new radiotherapy centre at the North West area in the United Kingdom.
NeuroSigma and the University of California (UCLA) sign agreement to develop...
The first two UCLA's thin-film nitinol (TFN) applications are flow-diverting stents for intracranial aneurysms and stents for treating peripheral artery disease.
Concentric Medical launches Dac 070, a new distal access catheter for...
The Dac 070 offers the stability of the original Dac catheters with the added benefit of a larger lumen through which physicians can deliver multiple therapeutic devices.
Texas Children’s Hospital pioneers use of MRI-guided laser surgery to treat...
According to hospital experts, this new surgical approach offers a safer and significantly less invasive alternative to craniotomy, currently the most commonly used cranial surgical treatment for epilepsy.
Envoy awarded Michael J Fox Foundation grant to develop improved oral...
Envoy Therapeutics' bacTRAP target discovery technology minimises activity in circuits that may trigger unwanted side effects such as dyskinesias, compulsive behaviors and somnolence, which are currently produced by Levodopa (the current gold standard in treating Parkinson's disease).
St Jude receives FDA approval for expansion of BROADEN deep brain...
The research focuses on Brodmann Area 25 region of the brain to help patients with major depressive disorder for whom current treatments are not effective.
Onyx works in the embolization of extracranial head and neck vascular...
New research has shown that Onyx (ev3/Covidien), known for its unique properties of being less adhesive and polymerising slowly, was able to stop acute bleeding in 13 out of 14 cases when used extracranially.
US medical data shows coiling has better outcomes than clipping for...
Fourteen per cent of patients who underwent surgical clipping were discharged to long-term facilities compared with 4.9% who underwent coiling. Clipped patients also had a higher mortality rate.
GE Global Research and Mayo Clinic awarded US$5.7 million to research...
Compared to equivalent field strength whole-body MRI scanner, GE's prototype scanner will be designed to deliver the same (or better) quality images, but from a scanner that is only one-third the size and significantly lighter.
NeuroTherm acquires ArthroCare’s Parallax and Contour product lines
Parallax and Contour are products used in interventional spine procedures. Parallax products have long been the standard for vertebroplasty and Contour has been developed for cavity creation.
Stryker expands interventional spine agreement with Novation
This agreement positions Stryker as the single source provider for kyphoplasty for VHA, UHC and Provista members.
Radiofrequency targeted vertebral augmentation shows lower cement leakage rate compared to...
A new study suggests that cement augmentation using radiofrequency targeted vertebral augmentation (RF-TVA) with the StabiliT system (DFine) may provide a lower leakage rate than vertebroplasty and balloon kyphoplasty in the treatment of vertebral compression fractures.
Andrew Platts is an editor-in-chief of NeuroNews
Dr Andrew Platts MB BS FRCS FRCR, consultant neuro and vascular radiologist with the Royal Free Hospital NHS Trust, London, UK, has just been appointed an editor-in-chief of NeuroNews.
CerebralRx launches FitNeS system for epilepsy treatment
FitNeS vagus nerve stimulation system is used to treat epilepsy patients with partial onset seizures who do not achieve full seizure control with available prescription drugs.
First patients in Australia received neurostimulation therapy with the Epiducer lead...
The Epiducer lead delivery system (St. Jude Medical) is designed to lessen procedural complexities and improve efficiency in the placement of neurostimulation leads utilised in spinal cord stimulation therapy for the management of chronic pain.
FDA approves first imaging agent to evaluate parkinsonian syndromes
DaTscan (GE Healthcare) is the first FDA-approved radiopharmaceutical adjunct imaging agent to help physicians evaluate patients with suspected parkinsonian syndromes, such as Parkinson's disease.
The neurobiological dimension of sexual orientation
"Sexual orientation is not a matter of choice, it is primarily neurobiological at birth", Jerome Goldstein, director of the San Francisco Clinical Research Center, USA, stated at the 21st Meeting of the European Neurological Society (ENS) in Lisbon.
Uniform distribution of endovascular coils may help in the treatment of...
US researchers found that the novel triangular primary wind design of Deltapaq endovascular microcoils (Codman Neurovascular) achieved a tighter packing density and a more uniform distribution of the coil mass across the aneurysm dome in comparison with helical and complex endovascular microcoils.
Neuralstem gets Safety Monitoring Board approval to advance spinal cord stem...
Neuralstem updated the progress of its ongoing phase I human clinical trial of the company's spinal cord stem cells in the treatment of ALS (amyotrophic lateral sclerosis, or Lou Gehrig's disease) at Emory University in Atlanta, Georgia, USA.
HydroCoil embolic system showed superior outcomes compared to bare platinum coils...
The HELPS trial results, published in The Lancet, showed that the HydroCoil system (MicroVention-Terumo) reduced recurrence and re-treatment rates and provided improved outcomes in patients when compared to bare platinum coils.
Penumbra system receives Shonin approval in Japan for use in the...
The Penumbra system (Penumbra) works on the proximal surface of the brain's vessel occlusion, optimising safety and eliminating the need for navigation beyond the occlusion.
Transcutaneous vagus nerve stimulation study shows promising results for patients with...
A decrease in the frequency and duration of hard to treat epileptic incidences was achieved for five of seven patients using transcutaneous vagus nerve stimulation at the Epilepsy Centre of the University Hospital Erlangen, Germany.
Doctors in Germany among first to use new self-expanding clot removal...
Revive SE (Codman and Shurtleff) is a self-expanding nitinol basket that provides a temporary bypass across the occlusion in the brain. It can also be used for the non-surgical removal of emboli and thrombi, and be used with aspiration.
Codman Neurovascular launches Orbit Galaxy detachable coil system
Orbit Galaxy features the first complete family of complex, random loop coils for the endovascular treatment of cerebral aneurysms, in the market.
New tool could help improve arm surgery for devastating nerve injury
Researchers at Hospital for Special Surgery (HSS), New York, USA, have developed a tool that may standardise international measure outcomes of different surgical treatments for brachial plexus injuries. This tool, if refined internationally, could help to improve arm surgery, investigators claimed.
Helicobacter pylori infection increases severity and frequency of migraine
Iranian researchers from Isfahan University of Medical Sciences and Islamic Azad University, Najafabd Branch, showed that Helicobacter pylori infections can affect one's head as well as stomach.
New study: post-stroke depression underestimated and undertreated
"Depression and anxiety are common after stroke, affecting one third of stroke survivors. Depression often goes unrecognised and untreated," explained Jennifer H White, University of Newcastle, Australia, at the 21st Meeting of the European Neurological Society (ENS) in Lisbon.
New therapy for restless leg syndrome in pregnant women shows positive...
A new study reveals iron substitution through infusions of ferric carboxymaltose as a potentially effective therapy for restless leg syndrome, one of the most common and bothering sleeping disorders in pregnant women.
Early Alzheimer related to cardiovascular risk factors such as hypertension or...
Patients with three or more cardiovascular risk factors seem to have a greater risk of developing symptoms of Alzheimer's disease before the age of 70 than patients with less than three cardiovascular risk factors.
One in seven strokes happen while sleeping
Approximately 14% of all strokes occur during sleep, preventing many from getting clot-busting treatment, according to a study published in the 10 May 2011, print issue of Neurology, the medical journal of the American Academy of Neurology.
FDA clears ReFlex catheter for distal intracranial access
The ReFlex catheter (Reverse Medical) is uniquely designed to provide endovascular intracranial access during interventional neurovascular procedures.
FDA approves new treatment for large brain aneurysms
The cPAX (Neurovasx) is indicated for use in aneurysms larger than 10 milimetres, up to now these aneurysms have been difficult to treat with clipping or coiling.
ReFlex A+ catheter receives CE mark approval for intracranial distal access...
ReFlex A+ (Reverse Medical) provides neurovascular specialists with a line of advanced access devices that can navigate into complex neurovascular anatomy, which may enable them to treat more patients than with currently available devices.
Uroplasty expands insurance coverage of posterior tibial nerve stimulation delivery in...
UnitedHealthcare and Excellus BlueCross BlueShield (BCBS) insurers initiated coverage of posterior tibial nerve stimulation effective from 1 April 2011 and 17 March 2011 respectively.
Silent strokes linked to acute anaemia in children
Silent strokes occur in a significant number of severely anaemic children, especially those with sickle cell disease, according to research presented at the American Stroke Association's International Stroke Conference 2011.
Gene therapy shows real promise in Parkinson’s
A multicentre gene therapy trial for patients with advanced Parkinson's disease demonstrated reduced symptoms of the progressive movement disorder, according to a new study published in Lancet Neurology.
FDA approves new medical device to treat glioblastoma multiforme
The NovoTTF-100A System is a non-invasive device, consisting of four sets of insulated electrodes attached to an electronic box that pumps low intensity electrical fields to stunt the glioblastoma multiforme tumor's growth.
Mini-stroke doubles risk of heart attack
Patients who have suffered a "mini stroke" or a transient-ischaemic attack (TIA) are at twice the risk of heart attack than the general population, according to research reported in Stroke: Journal of the American Heart Association.
Non-invasive brain stimulation may improve swallowing after stroke
Stroke patients who received transcranial direct current stimulation coupled with swallowing exercises showed greater improvement in swallowing ability than patients who did not receive this stimulation, according to a pilot study reported in Stroke: Journal of the American Heart Association.
New device uses submarine technology to diagnose stroke faster
A medical device developed by retired US Navy sonar experts, using submarine technology, is a new paradigm for the detection, diagnosis and monitoring of stroke, says a team of interventional radiologists at the Society of Interventional Radiology's 36th Annual Scientific Meeting, Chicago, USA.
Mayo Clinic receives gift award to further explore Lewy body dementia...
The Harry T Mangurian Jr Foundation's US$1 million gift will support studies designed to understand how Lewy body dementia develops, how to treat it more effectively, and how to diagnose it earlier.
Pipeline Embolization Device gets unanimous FDA Advisory Panel vote
Pipeline is intended for the endovascular treatment of large or giant wide-necked intracranial aneurysms in the cavernous and paraclinoid regions of the internal carotid artery. The FDA Advisory Panel recommended approval for the device.
Deep brain stimulation dramatically improves symptoms for patients with severe cervical...
Deep brain stimulation targeting the subthalamic nucleus, target widely used to treat Parkinson's disease, shows reduced pain and spasms without causing serious side effects for patients with severe cervical dystonia.
FDA Advisory Committee finds data support the safety of NovoTTF-100A System
The NovoTTF-100A is a portable, non-invasive medical device designed for continuous use throughout the day by patients with supra-tentorial glioblastoma multiforme tumours that recur after maximal surgical and radiation treatments.
FDA approves Benlysta to treat lupus
For the first time in over half a century, the FDA has approved a new drug to treat lupus. Benlysta is a monoclonal antibody, delivered directly by intravenous infusion that acts against B-lymphocyte stimulator.
Medtronic launches Activa SC deep brain stimulation system
Activa SC is used to treat the symptoms of advanced Parkinson's disease and essential tremor in the USA and Europe. The device is also approved for dystonia in Europe.
Device to maintain blood flow during artery bypass brain surgery gets...
The Elana surgical kit is an effective option for patients at greater risk for stroke during standard bypass surgery.
Stroke survivors with atrial fibrillation may have higher risk of dementia
Stroke survivors with atrial fibrillation were 2.4 times more likely to develop dementia than stroke survivors who did not have the heart condition, according to researchers from the University of East Anglia, Norfolk, UK.
BrainScope prepares to introduce Ahead EU-100 in the UK
The Ahead EU-100 device has the potential to represent a significant advancement in the early identification and assessment of patients with traumatic brain injury.
Clinical investigators address confusion over study of patent foramen ovale in...
Results in CLOSURE I study reinforce need for definitive study of patent foramen ovale in stroke patients.
Stroke in Mexican-Americans expected to rise 350% by 2050
Increases in strokes are expected to be considerably steeper in Mexican-Americans compared with non-Hispanic whites, according to research presented at the American Stroke Association's International Stroke Conference 2011.
Magnetic resonance imaging can help decide therapy in patients with unclear-onset...
Patients with unclear-onset strokes are usually excluded from clot-busting therapy because of lack of clarity on the beginning of their stroke symptoms; however research shows how magnetic resonance imaging can help to treat these patients safely.
tPA drug may improve outcomes in mild stroke patients
The intravenous tissue plasminogen activator (tPA), drug approved for severe ischaemic strokes, could also save US$200 million a year in disability costs in the USA.
Ischaemic stroke hospitalisations increase in young, research shows
Researchers are unclear on the reasons for the increase, but said obesity and hypertension may be factors.
Aneurysm embolization system receives CE mark
The Luna aneurysm embolization system (Nfocus Neuromedical) is designed to be a single-deployment, easy-to-use system for the treatment of brain aneurysms both pre- and post-rupture.
First patients enrolled in TREVO 2 clinical trial
TREVO 2 (Thrombectomy Revascularisation of large Vessel Occlusions) is a pivotal clinical trial investigating the safety and efficacy of the Trevo Retriever (Concentric) for removing clot from ischaemic stroke patients.
Most stroke patients are not getting clot-busting treatment in timely manner
Less than one-third of acute stroke patients receive plasminogen activator (tPA) within 60 minutes of their hospital arrival, according to research presented at the American Stroke Association's International Stroke Conference 2011.
Neuralstem receives FDA Orphan Drug Designation for spinal cord stem cells...
The NSI-566RSC is currently in phase I study to evaluate the safety of the product and the surgical route of administration to treat patients with amyotrophic lateral sclerosis.
Preliminary technology to detect Alzheimer’s disease uncovered
This technology could be used eventually in the development of specific biomarkers for Parkinson's disease, multiple sclerosis and lupus.
Young, uninsured or Medicare Part D survivors often cannot afford medicines...
Despite the US government prescription coverage, data suggest that medicine is still unaffordable for some disadvantaged stroke survivors. The results were presented by Deborah A. Levine at the American Stroke Association's International Stroke Conference 2011.
“Robot therapy adds a new wrinkle to stroke rehabilitation strategies”
Kayoko Takahashi, Japan, explained how therapy in which robots manipulate paralysed arms, combined with standard rehabilitation, can improve arm and shoulder mobility in patients after stroke.
Diet soda may raise odds of vascular events; salt linked to...
Studies presented at the American Stroke Association's International Stroke Conference 2011 showed that people who drank diet soda every day had a 61% higher risk of vascular events than those who reported no soda drinking. Also, people who consumed more than 4,000mg of sodium per day had more than double the risk of stroke compared to those consuming less than 1,500mg per day.
Advanced macular degeneration may be associated with an increased risk of...
The results of the Rotterdam study were presented by researchers from The Netherlands at the American Stroke Association's International Stroke Conference 2011.
Positron’s Attrius PET Scanner seeks market expansion into neurologic imaging
The conditional FDA approval of Eli Lilly's positron PET imaging agent, Amyvid, for detecting beta amyloid plaque deposits could help Positron's market expansion.
Ornim announces FDA market approval for non-invasive brain oximetry and blood...
The CerOx 3210F is currently the only device to use a single, non-invasive probe for simultaneously and independently measuring both oximetry and blood flow parameters, together providing a clear indication of tissue perfusion.
Serotonin levels can be detected and measured through an innovative device...
The Mayo Clinic study suggests that, in the future, measurements of serotonin may help establish a therapeutic mechanism of deep brain stimulation for psychiatric disease.
Could peripheral nerve field stimulation be an alternative to current pharmacological...
Peripheral nerve field stimulation is a safe, reversible, and effective treatment option for intractable headache and craniofacial pain, research presented at the 14th annual meeting of the North American Neuromodulation Society shows.
New Athena platform allows optimisation of deep brain stimulation therapy
The platform, produced by St. Jude Medical, has recently received regulatory approval from the Australian Therapeutic Goods Administration (TGA).
New eye-tracking device for earlier diagnosis of Parkinson-plus diseases
Results have shown that eye movements provide a more accurate early diagnosis than traditional clinical examinations.
BrainScope announces sponsorship to multicentre clinical study of head injury in...
Ahead (BrainScope) medical device in development will be used to collect, process and produce an assessment of the patients' brain electrical activity, identifying patterns consistent with a traumatically-induced structural brain injury.
Deep brain stimulation is an effective therapy for advanced Parkinson’s disease
This procedure does not replace drugs but it often allows their dosage to be reduced; the combination provides better muscle and movement control than drugs alone, according to research published in the International Journal of Neuroscience.
Coma and general anaesthesia demonstrate important similarities
The New England Journal of Medicine review into brain circuit mechanisms may lead to more accurate coma diagnosis and improved therapies.
Complications of deep brain stimulation
A new study shows that the highest risks associated with primary deep brain stimulation implants are those related to the implants rather than to the actual procedure of their placement in the brain.
New 18F-flutemetamol data for detection of amyloid plaque linked to Alzheimer’s...
18F-flutemetamol is currently in phase III of development and is being studied via imaging of the brain tissue in live humans.
Boston Scientific acquires Intelect Medical
Intelect Medical develops innovative technology designed to advance deep brain stimulation therapy.
Large stroke genetics research project gets funding
US$3.6 million grant from National Institutes of Health to fund research at 17 sites in USA and Europe to uncover genetic influences for prevention and treatment of stroke.
Stryker completes acquisition of Boston Scientific’s Neurovascular division
Stryker Corporation has announced the closing of its acquisition of the assets of the Neurovascular division of Boston Scientific which includes products used for the minimally invasive treatment of haemorrhagic and ischaemic stroke.
Neuronetrix begins pivotal multicentre clinical trial of Alzheimer’s diagnostic device
Neuronetrix has announced the first test of the Cognision System as part of the company’s pivotal multicentre clinical trial.
Teva announces successful results of phase III study with oral Laquinimod...
Laquinimod study met primary endpoint of reducing annualised relapse rate. Treatment with the drug significantly slowed progression of disability and the data showed a favourable safety and tolerability profile.
St. Jude Medical neurostimulation study demonstrates sustained improvement in chronic low...
Results presented at the 14th annual North American Neuromodulation Society meeting show that 70% of patients report overall pain relief
Inspire Medical Systems announces FDA approval for STAR trial
Inspire neurostimulation device for obstructive sleep apnea will be tested at sites in US and Europe.
St Jude Medical announces results of ASSERT study demonstrating effectiveness of...
These results are significant because they demonstrate that even brief, asymptomatic atrial fibrillation episodes can cause a stroke, and that the proportion of strokes that are associated with atrial arrhythmias is much higher than previously thought.
Boston Scientific begins patient enrollment in clinical trial assessing deep brain...
Company implants first deep brain stimulation device designed to treat Parkinson's.
Siemens presents a new consulting concept for optimised stroke care in...
Siemens sector Healthcare has developed a new consulting model "Act on Stroke", tailored specifically to clinical workflows in the treatment of stroke.
Study suggests physicians wait longer for signs of brain recovery after...
Experts at Johns Hopkins say that physicians might be drawing conclusions too soon about irreversible brain damage in patients surviving cardiac arrest whose bodies were for a day initially chilled into a calming coma.
Boston Scientific agrees to sell Neurovascular business to Stryker
Boston Scientific Corporation has announced the execution of a definitive agreement under which Stryker Corporation will acquire Boston Scientific’s Neurovascular business.
St. Jude Medical announces approval of the first rechargeable neurostimulator to...
The Eon Mini spinal cord stimulator is the world's smallest, longest-lasting rechargeable neurostimulator to manage chronic pain, combining greater patient comfort with a 10-year battery longevity.
Study supports the long-term benefits of TMS for depression
In a study to determine the durability and long-term effects of TMS, psychiatric researchers at Rush University Medical Centre have found the non-invasive, non-drug therapy to be an effective, long-term treatment for major depression. Results of the study were published in the October 2010 issue of Brain Stimulation.
Study could pave way for more effective Alzheimer’s treatments
Scientists have determined that a new instrument known as PIB-PET is effective in detecting deposits of amyloid-beta protein plaques in the brains of living people, and that these deposits can predict who will develop Alzheimer’s disease
Neuroimaging: new insights into MS, ALS, dementia and migraine
Innovative imaging techniques such as functional MRI or diffusion tensor imaging occupy an important place in modern neurology today by helping to get a better understanding of diseases including multiple sclerosis, dementia, amyotrophic lateral sclerosis and Alzheimer's disease
Stem cells to help study of Parkinson’s disease
A new technology is to be used by Oxford University researchers to better understand the causes of Parkinson's disease
Sleep disorder may help predict neurological disease
Idiopathic REM sleep behaviour disorder may be the initial manifestation of synucleinopathies
Gene identification “a step forward”
Parkinson's disease sufferers have been offered new hope after the gene mutation which triggers it was identified by scientists
New drugs reduce attacks in MS and improve walking ability
Cladribine and fingolimod which can be taken in pill form, as a short course treatment with time dependent effect, or as a daily dosage, significantly reduce the frequency of attacks of multiple sclerosis.
Botox eases nerve pain
Botox, a toxin known to weaken or paralyse certain nerves and muscles, may have another use that goes beyond the cosmetic.
Is venous stenting really an option for multiple sclerosis?
A brand new theory, the liberation theory which proposes that angioplasty or venous stenting could relieve some of the symptoms of MS, and that concomitant to revolutionary theories, a huge dollop of scepticism.
Positive initial results for DBS in Alzheimer’s
Insertion of a deep brain stimulator (DBS) may improve memory and function for patients with Alzheimer's disease according to a new study published in Annals of Neurology
St. Jude medical announces European approval of next generation Athena programmer...
Easy to use programmer allows clinicians to optimise deep brain stimulation therapy through advanced programming and data management features.
Teriflunomide significantly reduces annualised relapse rate and was well tolerated in...
Sanofi-aventis announces that teriflunomide significantly reduces annualised relapse rate at 2 years versus placebo in patients with relapsing multiple sclerosis.
Neuralstem files FDA application for first chronic spinal cord injury stem...
Multicentre phase I safety trial will enroll "complete paralysis" patients.
A promising target for developing treatments against Parkinson’s
Results in mice offer a breakthrough in treatment of patients with LRRK2 mutations.
Alzheimer’s imaging study identifies changes in brain’s white matter
Data suggests decreased integrity in white matter connections may be the one of the earliest brain changes in the disease.
FDA approval of new leads for Boston Scientific Precision Plus Spinal...
Company now offers pain specialists the most comprehensive portfolio of SCS percutaneous leads.
Medtronic launches novel clinical trial evaluating Spinal Cord Stimulation Therapy for...
Industry first Defeat-HF Trial aims to slow HF progression with neurostimulation.
New guideline: MRI better than CT scans at diagnosing stroke
One study shows stroke accurately detected 83% of the time by MRI, versus 26% by CT.
Longer post-stroke time frame for thrombolysis
Large scale European study shows thrombolysis is still effective up to four and a half hours after a stroke.
3D eye imaging enters neurology offices
Heidelberg Engineering presented axonal tracking technology recently
New guidelines on brain death diagnosis
The American Academy of Neurology has issued new guidelines for determining brain death. These have been updated for the first time in 15 years and provide step-by-step instructions to help guide physicians through the complex process of determining brain death in adults.
Florbetaben may help visualise early stages of Alzheimer’s disease
A multinational clinical trial presented at Society of Nuclear Medicine's 57th annual meeting notes that a novel imaging agent, florbetaben, could be the next major breakthrough for the early detection of Alzheimer's disease.
Medtronic announces neurostimulation therapy clinical trial for intractable chronic pain
Medtronic has announced the initiation of a prospective, multicentre, randomised USA clinical trial for the RestoreSensor neurostimulator that adapts therapy to the changing needs of chronic pain patients by responding automatically to changes in body position and activity in an upright position.
St Jude Medical’s Epiducer lead delivery system gets CE mark
St Jude Medical has announced European CE Mark approval of its new Epiducer lead delivery system for neurostimulation therapy, the first of its kind, allowing introduction of S-Series perc-paddle leads as well as multiple lead arrays through a single percutaneous entry point.