Initial experience with PulseRider (Pulsar Vascular) has shown the device to be safe and effective as an adjunct in the treatment of bifurcation aneurysms arising at the basilar apex or carotid terminus according to a report of the first three cases in the USA published in the Journal of NeuroInterventional Surgery.
In June of 2014, the US Food and Drug Administration (FDA) approved an investigational device exemption (IDE) for the PulseRider, allowing Pulsar Vascular to begin a multicentre clinical trial in support of a humanitarian device exemption to evaluate the PulseRider for US approval for wide neck aneurysms at or near a bifurcation of the basilar tip or carotid terminus. PulseRider is currently only available in the USA in the context of the Adjunctive neurovascular support of wide-neck aneurysm embolization and reconstruction (ANSWER) clinical trial.
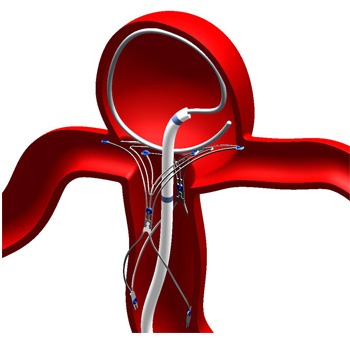
The first three cases were done by physicians from the Medical University of South Carolina, Charleston, USA. Alejandro Spiotta et al explain that patients were pretreated the evening before the procedure with clopidogrel and aspirin and then maintained on daily doses thereafter. All cases were performed under general endotracheal anaesthesia. An appropriately sized PulseRider device was deployed across the neck of the aneurysm. A microcatheter was then navigated over a 0.014 inch microwire through the device into the aneurysm. In these first three cases, complete occlusion of the aneurysm was achieved without intraprocedural complications.
The authors report: “In our early experience we have found [PulseRider’s] use to be simple and safe, being readily delivered in a standard method very similar to other available stents, making the procedure more familiar to an operator new to the device.”
Confirming the safety and effectiveness of the device in their first experiences, they conclude that the PulseRider “represents a useful addition to the armamentarium of the neuroendovascular specialist”.













