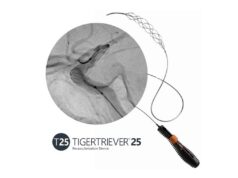
Rapid Medical has announced the US Food and Drug Administration (FDA) clearance of its Tigertriever revascularisation device for use in the treatment of ischaemic stroke. According to a company press release, this is the world’s first adjustable thrombectomy device.
Rapid Medical has announced the US Food and Drug Administration (FDA) clearance of its Tigertriever revascularisation device for use in the treatment of ischaemic stroke. According to a company press release, this is the world’s first adjustable thrombectomy device.
Rapid Medical claims that the Tigertriever is the first stent retriever to offer intelligent control, enabling neuro interventionalists to better remove blood clots and restore blood flow to the brain following an ischaemic event.
This clearance comes after the results from the 160-patient TIGER (Treatment with the intent to generate endovascular reperfusion) trial were presented at the International Stroke Conference (ISC; 17–19 March, virtual). Rapid Medical report that the TIGER trial demonstrates a statistically significant improvement in restoration of blood flow to the brain during ischaemic stroke and equivalent safety for the Tigertriever device, compared to existing self-expanding stent retrievers.
“The trial confirmed that the Tigertriever is not only safe and effective, but demonstrably improved outcomes over similar trials testing previous technology,” observed Jeffery Saver, professor of Neurology and Director of the Comprehensive Stroke and Vascular Neurology Programme at University of California, Los Angeles, Los Angeles, USA. “Tigertriever is an important advance in stroke treatment devices and a win for patients.”
“In the TIGER trial, we saw an 11-point improvement in revascularisation with Tigertriever over the data from recent stent retriever trials. This may improve outcomes for one in 10 stroke patients, a major leap from previous devices,” stated Rishi Gupta, national co-principal investigator of the TIGER trial, vascular neurologist and director of Neurocritical Care at WellStar Health System in Marietta, USA. “I have more control over the clot when I need it,” added Gupta.
The trial demonstrates 84.6% revascularisation success, compared to a 73.4% average from six recent pivotal stent retriever trials. Additionally, 58% of patients were living independently after 90 days versus an average of 43% of patients from the previous device trials. Furthermore, the TIGER trial’s safety outcomes showed an 18.1% primary safety score versus a 20.4% historical average.
“Tigertreiver’s intelligent control represents a new generation of stent retrievers as shown by the highest revascularisation success rate of similar stent retriever trials,” said Walid Haddad, chief clinical officer at Rapid Medical.
Ronen Eckhouse, Rapid Medical co-founder and CEO states, “The FDA clearance of Tigertriever is a significant milestone in the development and growth of Rapid’s global business. It is a great testament to the advantages of our proprietary 3D braiding technology—the platform for our portfolio of remotely adjustable neurovascular devices, including the Comaneci and Columbus products.”
Tigertriever already has its CE mark and is available in the USA in 21mm and 17mm sizes.