NeuroOne Medical Technologies has announced that it has received US Food and Drug Administration (FDA) 510(k) clearance to market its Evo stereoelectroencephalography (sEEG) electrode technology for temporary (less than 30 days) use with recording, monitoring and stimulation equipment for the recording, monitoring, and stimulation, of electrical signals at the subsurface level of the brain.
In August this year, the company announced it had resubmitted its 510(k) application to the FDA for less-than-30-day use, which included additional biocompatibility testing as requested by the FDA. And, on 20 October, it received an FDA clearance letter stating that the Evo sEEG system is substantially equivalent to the predicate device and may be marketed in the USA.
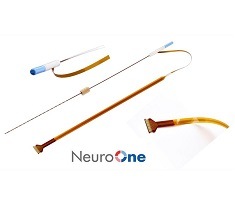
The Evo sEEG system represents NeuroOne’s second FDA 510(k) cleared product. As opposed to cortical electrodes, sEEG electrodes provide a similar function at the subsurface level of the brain by using a much less invasive process that does not require removal of the top portion of the patient’s skull, a company press release notes.
NeuroOne’s Evo Cortical and sEEG electrodes are a portfolio of high-definition, thin-film electrodes. The release also states that potential advantages include increased signal clarity and reduced noise; better tactile feedback during insertion into brain tissue; and faster order fulfilment due to an automated manufacturing process.
As previously reported, NeuroOne is also advancing a pipeline of therapeutic electrode technologies for brain-tissue ablation, and chronic stimulation use for deep brain stimulation (DBS) and spinal cord stimulation (SCS) for chronic back pain.