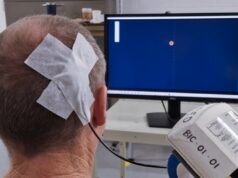
In patients with treatment-resistant depression (TRD), multiple sessions of intermittent theta-burst stimulation (iTBS) do not accelerate responses to the therapy, nor do they improve depressive symptoms. This is the key finding of the CARTBIND (Canadian rTMS treatment and biomarker network in depression) study—a randomised, sham-controlled comparison of once- versus twice-daily iTBS for depression published in the journal Brain Stimulation.
In patients with treatment-resistant depression (TRD), multiple sessions of intermittent theta-burst stimulation (iTBS) do not accelerate responses to the therapy, nor do they improve depressive symptoms. This is the key finding of the CARTBIND (Canadian rTMS treatment and biomarker network in depression) study—a randomised, sham-controlled comparison of once- versus twice-daily iTBS for depression published in the journal Brain Stimulation.
The study, authored by Daniel Blumberger (University of Toronto, Toronto, Canada) and colleagues, highlights the urgent need for more scalable and tolerable treatment methods for patients suffering from TRD. The authors write that repetitive transcranial magnetic stimulation (rTMS) is an established, evidence-based treatment for TRD, but that iTBS can be administered in one tenth of the time of rTMS (~3 minutes vs 37.5 minutes) yet achieves similar or greater effects on neural plasticity and depressive symptoms. Therefore, Blumberger et al set out to investigate the impact of multiple iTBS sessions per day in patients with TRD.
Patients were recruited from across three Canadian academic health sciences centres—the Centre for Addiction and Mental Health and the University Health Network, both of which are affiliated with the University of Toronto, and the University of British Columbia Hospital—between April 2016 and February 2018. To be eligible, participants had to fall between the ages of 18 and 59 years, and have a confirmed diagnosis of major depressive disorder.
Additionally, patients had to meet the following criteria: failed to achieve a clinical response to an adequate dose of an antidepressant based on an antidepressant treatment history form (ATHF) score ≥3 in the current episode or were unable to tolerate two separate trials of inadequate dose and duration; had a score ≥18 on the Hamilton Depression Rating Scale (HRSD-17); had no increase or initiation of any psychotropic medication in the four weeks prior to screening; were able to adhere to the schedule; passed the adult safety screening (TASS) questionnaire, and had normal thyroid functioning based on pre-study blood work.
The primary outcome measure was changes in HRSD-17, from baseline to day 10 of treatment, with the secondary outcome measure being the change from baseline to day 30 of treatment. In this study, remission was defined as a HRSD-17 score of less than eight and the response was defined as a decrease in HRSD-17 score of 50% or less. These outcome measures were analysed at both day 10 and day 30 of treatment.
Blumberger et al assessed a total of 265 patients, of which 208 were eligible for the study. These patients were randomised to either receive once-daily iTBS with one sham treatment (n=105; control, one-time 1200 pulses daily group) or twice-daily iTBS (n=103; accelerated, two-time 600 pulses daily group). In the once-daily group (60% female) and twice-daily group (66% female), the average ages were 41.2±11.5 years and 40.7±11.1 years, respectively. The age of depression onset was earlier in the twice-daily group at 20.7±10.5 years compared to 21.9±10.2 years in the once-daily group. Additionally, the baseline HRSD-17 was 23.2±4.0 in the twice-daily group and 23.1±3.8 in the once-daily group.
A total of 198 participants completed the treatment on day 10, with 185 completing six weeks of treatment. At one, four and 12 weeks, 175, 157, and 141 participants received follow-up, respectively. The results revealed no significant difference in changes in the HRSD-17 scores from baseline to day 10 between the two groups (p=0.84)—however, a significant difference was seen in both the twice-daily (estimated adjusted difference -6.46 points, p<0.001) and once-daily (estimated adjusted difference -6.66 points, p<0.001) groups individually. Furthermore, there were no significant differences in changes in HRSD-17 between the groups from baseline to day 30 (p<0.001), although a significant change in HRSD-17 scores from baseline to day 30 for both the twice-daily group (estimated adjusted difference -10.52 points, p<0.001) and the one-daily group (estimated adjusted difference -9.05 points, p<0.001) was observed.
The authors note that there are several limitations to this study that may have affected the outcomes. The 30-day length of treatment may have led to an expectancy effect and negatively biased early treatment effects, while the doubling of sessions resulted in prompting for—and as such potential over-reporting of—side-effects, Blumberger et al write.
The authors conclude that, while it is important to consider patient expectations in comparisons of brain stimulation techniques such as this, remission and responses rates did not differ between the two study groups, and an acceleration in response was not observed when controlling for number of pulses and operator contact. As such, the study’s concluding findings ultimately stress that doubling sessions of iTBS does not yield a more rapid reduction in depressive symptoms, nor does it lead to improved outcomes in TRD patients.