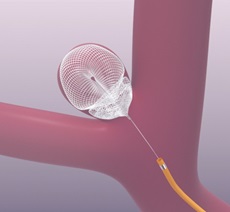
Galaxy Therapeutics has announced the submission of the final module of its premarket approval (PMA) application to the US Food and Drug Administration (FDA) for the Seal device for the treatment of wide-neck bifurcation aneurysms (WNBAs).
This modular PMA submission is supported by data from the WNBA cohort of the SEAL IT investigational device exemption (IDE) trial, which enrolled 163 patients across leading US neurointerventional centres. The submission includes the WNBA cohort only, Galaxy notes in a press release.
SEAL IT was designed to evaluate the Seal device in a real-world patient population, including a broad range of aneurysm sizes and anatomical complexities, the company adds, also stating that the study represents “one of the most comprehensive prospective US studies in intracranial aneurysms”, with dedicated cohorts evaluating wide-neck bifurcation, sidewall and ruptured aneurysms.
“Submitting the final PMA module for Seal marks a major milestone in our journey toward bringing this technology to patients,” said Osama Zaidat (Mercy Hospital, Toledo, USA), president and chief executive officer (CEO) of Galaxy. “This achievement reflects the strength of our clinical programme and our commitment to advancing the treatment of complex brain aneurysms.”
“This submission is the result of strong execution across clinical, manufacturing and regulatory functions,” added Jim Twitchell, chief operating officer at Galaxy. “We have built the operational foundation required to support the next phase of growth and future commercialisation.”
Galaxy also notes that it will continue to collaborate with the US FDA during the PMA review process.










