Tag: SEAL IT
Galaxy submits final PMA module for Seal device in wide-neck bifurcation...
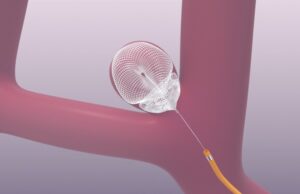
Galaxy Therapeutics has announced the submission of the final module of its premarket approval (PMA) application to the US Food and Drug Administration (FDA)...
Galaxy completes enrolment in pivotal trial evaluating Seal intrasaccular device
Galaxy Therapeutics has announced it has successfully completed enrolment in SEAL IT, the company’s US investigational device exemption (IDE) pivotal trial evaluating the Seal...
Galaxy Therapeutics completes enrolment of primary cohort in pivotal SEAL IT...
Galaxy Therapeutics has today announced completed enrolment of the primary cohort of patients in its SEAL IT investigational device exemption (IDE) trial.
The trial's primary...