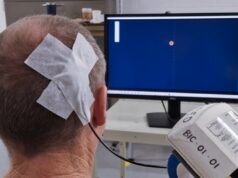
The European Society for Brain Stimulation (ESBS) has said it “strongly opposes” the European Union’s (EU) recent decision to reclassify non-invasive brain stimulation (NIBS) technologies as Class III medical devices.
In what ESBS members describe as a “sudden and unwelcome” regulatory change, which was announced in December 2022, repetitive transcranial magnetic stimulation (rTMS) and low-intensity transcranial electrical stimulation (tES) devices are now set to be considered in the highest-risk category under the EU Medical Device Regulation (MDR).
“Although this new reclassification currently refers only to ‘products without an intended medical purpose’ (a terminology that is, for many researchers and medical doctors, unclear), the evidence adduced about the risks and adverse effects of rTMS and tES—which serves as the justification for this reclassification—is gravely flawed,” Andrea Antal (University Medical Center Göttingen, Göttingen, Germany) et al write in Brain Stimulation.
While the authors note their agreement that all NIBS devices must be certified as medical devices—something that was not extensively defined within the previous Medical Device Directive (MDD)—they argue a more moderate-risk Class IIa classification would be more appropriate.
“To reclassify NIBS devices as having the same level of risk as invasive brain stimulation devices that are implanted inside the brain is inappropriate, contradicts 30 years of safety data, and has been decided without consultation of relevant professional stakeholders,” they add.
According to the ESBS, the EU’s assessment that NIBS devices pose a greater risk to patient safety than previously thought is based on “incorrect statements” about rTMS and low-intensity tES, citing claims and assumptions that are “simply false”—such as TMS/tES inducing atypical brain development and abnormal patterns of brain activity—and concerns around rTMS/tES-related seizure risks that are “no longer supported by scientific evidence”.
The current scientific and clinical evidence suggests both rTMS and low-intensity tES are safe treatment and research interventions, with few and mild adverse effects, making the establishment of this new EU ruling “hard to understand”, in the authors’ view.
NIBS products previously listed as Class I or IIa will be allowed to remain on the market—provided they comply with EU transition rules—until the MDR deadline, which was recently extended from May 2024 to December 2027 for higher-risk devices, and December 2028 for medium- and lower-risk devices.
Nevertheless, the ESBS states a belief that this reclassification will “have significant negative consequences for the future of our field”, resulting in higher costs and substantial research and development delays in the short term; and ultimately making NIBS treatments less accessible for European patients in the long run.
“European citizens will be disadvantaged, and there is the risk that other treatment approaches with more serious and established adverse effects will be overused to compensate for the lack of NIBS availability,” Antal and colleagues conclude. “Consequently, we strongly protest this decision, and we urge our colleagues working in our field to do the same, regardless of nationality. We have already sent a protest letter to the EU.”