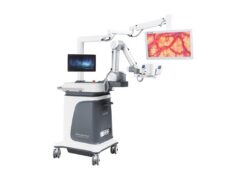
Aesculap has announced its receipt of 510(k) clearance from the US Food and Drug Administration (FDA) for DIR800 3D digital infrared fluorescence for use with its Aesculap Aeos robotic digital microscope. In a company press release, Aesculap report clearance further enhances the fluorescence capabilities of the microscope which already includes DUV400 3D digital ultraviolet mode.
According to Aesculap, the DIR800 is an accessory for the Aesculap Aeos robotic digital microscope and is used in viewing intraoperative blood flow in the cerebral vascular area including, but not limited to, assessing cerebral aneurysm and vessel branch occlusion, as well as patency in neurosurgery. The company further states that it also aids in the visual assessment of intraoperative blood flow and vessel patency in bypass neurosurgical procedures.
The press release reports that with DIR800, the Aesculap Aeos robotic digital microscope produces excitation light to illuminate the fluorescence properties of Indocyanine Green (ICG). The generated fluorescence signal depicts the distribution of the infrared dye in the patient’s blood vessels during surgery.
Aesculap claims DIR800 combines infrared fluorescence technology with 3D digital visualisation while also allowing the surgeon and operating room team to share the same high-quality live view and slow motion playback on multiple 3D 4K monitors. Backlight illumination aids visualisation of nearby non-fluorescing structures without the need to switch back to white light.
Finally, Aesculap reports another key benefit is heads-up positioning to free the surgeon from the microscope ocular tether, allowing the surgeon, not the microscope, to dictate ergonomics.