Better understanding of the brain alongside the integration of artificial intelligence may represent the future of neuromodulation. From machine learning and novel technology to non-invasive brain-computer interfaces that may hold the potential to repair brain damage; Dirk De Ridder (University of Otago, Dunedin, New Zealand) speculates what the development of this field permits, as well as what has been holding it back.
The treatment of neurological and psychiatric disease is compromised by the recent (+/- 2011) disengagement of the big pharma in developing novel medications for brain disorders. The reason is that designing drugs for central nervous system disorders has 50% less chance of making it to market and takes 30% longer than for other indications, such as cancer and heart disease.1,2
Thus, a novel non-pharmacological way of treating brain related pathologies is of crucial importance if progress is to be made in treating these most debilitating disorders. Interestingly, this methodology is already available, albeit in an immature fashion.
More than 60 years ago, in 1952, Delgado described implantation of intracranial electrodes for recording electrical brain activity as a diagnostic tool, and deep brain stimulation via the same electrodes as a possible treatment for mental disorders.3 This concept was based on the clinically beneficial effects of destructive psychosurgery through lesioning and the then recent (1947) development of stereotaxy for better targeting.4 The described technique became adapted for movement disorders in 1963 by Bechtereva in Russia5 and later in 1987 by Benabid in the Western world.6 The last 30 years has however been characterised by a stagnation in novel technology for brain stimulation, in sharp contrast to the exponential technological advancement in consumer devices such as smart phones, personal computers, etc.
If neuromodulation is to fill in the gap left by the big pharma’s withdrawal from neuroscience it must mature to a sophistication that goes beyond technology from the last millennium. But what are the essential ingredients for this evolutionary jump in neuromodulation technology to occur for the benefit of desperate patients with unaided brain disorders, whether neurological or psychiatric?
The future of neuromodulation fundamentally depends on three pillars: first; better understanding of the brain, second; better technology, and lastly; integration of artificial intelligence (AI) for the development of smart neuromodulation.
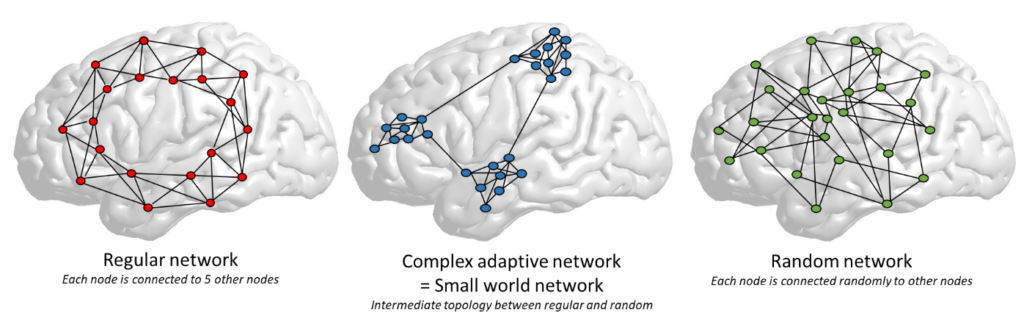
Largely due to the impetus given by the ‘decade of the brain (1990-2000)’, the understanding of the brain morphology, physiology and pathophysiology has increased dramatically, yet this has not yet been implemented in brain stimulation for clinical purposes. The brain is currently seen as a complex adaptive system, defined by the presence of a small world topology and embedded noise.7 Small world topology means that the structure and function of the brain is intermediate between two extremes, a fully deterministic regular or lattice network on the one hand, and a completely chaotic unpredictable8 random topology on the other hand. A small world topology permits adaptive flexibility, which neither of the two extremes exhibit (figure 1).
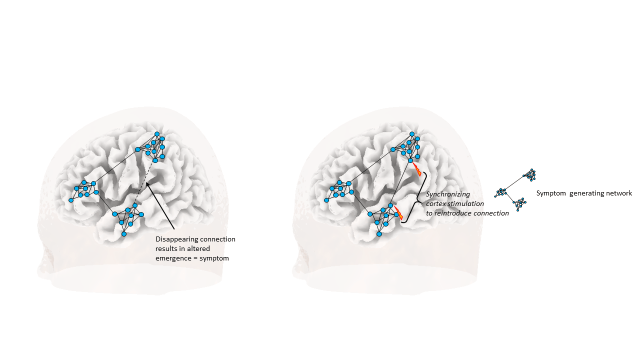
All complex adaptive systems, whether economy, internet, ant colony or brain. exhibit the same characteristics, in that they are complex, and adaptive—they have the capacity to change and learn from experience. They also show self-organisation, in that their complexity increases without an external organiser, as well as self-similarity, meaning that the whole has the same shape as one or more of the parts. But most importantly they are characterised by emergence. Emergence arises when an entity has properties its parts do not have, due to interactions between the parts. As such any symptom or brain disorder can be seen as an emergent property of a reorganized small world network.9 Neuromodulation can therefore be seen as a way to normalise symptom-generating reorganized networks (figure 2).
This implies that the future of neuromodulation will have to incorporate the neurophilosophy of network science if it is to mature to a treatment worthy of the third millennium, analogous to what is seen in the development of self-driving cars and so forth.
This better understanding of the brain as a complex adaptive small world system requires better technology, not just for the deployment of novel stimulation designs(10) such as burst stimulation,11 but also by incorporating sensing technology, which permits to record brain activity and connectivity measures, analogous to and extending from what is already available in pacemaker technology. The first initial steps are being taken by the industry for feedback-based, closed-loop spinal cord stimulation in the treatment of pain,12 and responsive brain stimulation for epilepsy,13 demonstrating the potential of this technology. Yet, these closed-loop and responsive stimulations are not smart in that they don’t learn (figure 3 left).

These systems behave like a servo-system in a car, without prediction, and cannot be applied beyond what they are programmed for. A smart system can learn to better detect the neural activity and connectivity signatures of the presence of a symptom as well as the intensity of a symptom, whether sensory, motor, cognitive, emotional or social. A smart system can learn to select an optimal stimulation design to better normalise the symptom by increasing or decreasing connectivity and thus changing the network structure. And optimally, a smart system can predict symptoms as to prevent their re-occurence in chronic disease states. Furthermore, a smart system improves itself, and learns to respond differentially depending on the context and intention (figure 3 right).
In order for neuromodulation to become smart, it needs to embed AI in its design, which controls both the sensing and execution of the neuromodulation device. In essence, the device needs a little brain to control itself, in order to become flexible and adaptive, in order to learn from the past to predict how it should behave in the future in a similar context depending on a specific goal. This tends to frighten many people, who fear computers will take over control from humans, once AI supersedes human intelligence, in a moment coined the singularity, predicted to occur around 2045.14
So, how far are we? What is the current state of affairs? Is there novel technology that could be useful for the development of smart neuromodulation? And, is there interest from the industry in the development of flexible adaptive brain-computer interfaces?
A first step in adaptive flexible neuromodulation is the decoding of symptoms, thoughts, memories, actions, perceptions, and so on. Recently, AI using machine learning has been capable of finding an objective signature for symptoms such as pain, tinnitus, tremor, depression,15 but also for memories,16,17 which can even be used for restoring memories and even transplanting them in other animals.18
But once sensory, emotional, cognitive or social stimuli are detected they need to lead to a percept, i.e. the interpretation of a stimulus.19 Electrical microstimulation of the somatosensory cortex for example with high resolution high density electrodes permits to induce different sensory percepts that feel fairly natural.20 It is clear that neuroscientific knowledge is currently only at the initial stage of cognitive, emotional and social decoding and of artificial sensory, thought or memory perception generation, but with the very rapid growth of machine learning, decoding will be rapidly improving. Yet, at present, high resolution stimulation is required and is still running behind.
In order to sense and stimulate novel, less invasive electrode designs with improved integration in the brain is also required. This could be aided by novel technologies such as syringe injectable mesh electronics,21 which in animals are shown to integrate seamlessly with brain in contrast to thin film electronics22 and result in stable chronic recording of brain activity.23 Neural dust, 1mm sized individual electrodes use ultrasonic power and communication to enable a scalable, wireless, and battery-less system for interfacing with the nervous system.24
These new technologies combined with greatly improved artificial intelligence will permit in the future to construct invasive and non-invasive brain-computer interfaces, that could integrate with brain tissue and thus repair brain damage, normalise pathological activity and connectivity or even enhance normal brain functioning. This has led to a race between non-neuromodulation companies such as google, facebook, DARPA, Kernel, Neuralink, Softbank vision fund to develop an ultra-fast high bandwidth brain-computer interface. Visionaries such as Elon Musk (Neuralink) dream to extend Paul MacLean’s triune brain25 with electronics. The underlying idea is that Paul MacLean’s vision of the brain as a structure with three evolutionary layers (the brainstem being the reptilian brain, the cingulate cortex defined as the emotional brain, and the cortex dedicated to cognition) will be extended with a fourth layer of electronics that will permit for each individual brain to become linked to the cloud.
Whereas for now this is clearly impossible, visions such as these combined with the singularity idea of AI surpassing all of human intelligence pave the way for futurologists and science fictionists to envision a future in which the boundaries between humanity and computers become progressively more blurred. Whether we want this or not, this avenue is the way neuroscience currently moves, and the medical neuromodulation field can only benefit from these evolutions. The industry has understood this and large companies such as Abbott are hiring people from google and other companies who push the limits of AI to drive innovation in neuromodulation.
In conclusion, the future of neurostimulation is smart neuromodulation.
Dirk De Ridder is a Belgian neurosurgeon. He is currently the Neurological Foundation Professor of Neurosurgery at the University of Otago in Dunedin, New Zealand.
References:
- Gribkoff VK & Kaczmarek LK (2017) The need for new approaches in CNS drug discovery: Why drugs have failed, and what can be done to improve outcomes. Neuropharmacology 120:11-19.
- Kesselheim AS, Hwang TJ, & Franklin JM (2015) Two decades of new drug development for central nervous system disorders. Nat Rev Drug Discov 14(12):815-816.
- Delgado JM, Hamlin H, & Chapman WP (1952) Technique of intracranial electrode implacement for recording and stimulation and its possible therapeutic value in psychotic patients. Confinia neurologica 12(5-6):315-319.
- De Ridder D (2013) A short history of neurosurgical localization. World Neurosurg 80(5):479-481.
- Bechtereva NP, Bondartchuk AN, Smirnov VM, Meliutcheva LA, & Shandurina AN (1975) Method of electrostimulation of the deep brain structures in treatment of some chronic diseases. Confinia neurologica 37(1-3):136-140.
- Benabid AL, Pollak P, Louveau A, Henry S, & de Rougemont J (1987) Combined (thalamotomy and stimulation) stereotactic surgery of the VIM thalamic nucleus for bilateral Parkinson disease. Applied neurophysiology 50(1-6):344-346.
- Amaral LA, Diaz-Guilera A, Moreira AA, Goldberger AL, & Lipsitz LA (2004) Emergence of complex dynamics in a simple model of signaling networks. Proc Natl Acad Sci U S A 101(44):15551-15555.
- Bullmore E & Sporns O (2009) Complex brain networks: graph theoretical analysis of structural and functional systems. Nat Rev Neurosci 10(3):186-198.
- Barabasi AL, Gulbahce N, & Loscalzo J (2011) Network medicine: a network-based approach to human disease. Nat Rev Genet 12(1):56-68.
- De Ridder D, Perera S, & Vanneste S (2017) State of the Art: Novel Applications for Cortical Stimulation. Neuromodulation 20(3):206-214.
- De Ridder D, Vanneste S, Plazier M, & Vancamp T (2015) Mimicking the brain: evaluation of St Jude Medical’s Prodigy Chronic Pain System with Burst Technology. Expert Rev Med Devices 12(2):143-150.
- Russo M, et al. (2018) Effective Relief of Pain and Associated Symptoms With Closed-Loop Spinal Cord Stimulation System: Preliminary Results of the Avalon Study. Neuromodulation 21(1):38-47.
- Geller EB (2018) Responsive neurostimulation: Review of clinical trials and insights into focal epilepsy. Epilepsy Behav.
- Kurzweil R (2005) The Singularity is Near (Penguin, New York).
- Vanneste S, Song JJ, & De Ridder D (2018) Thalamocortical dysrhythmia detected by machine learning. Nature communications 9(1):1103.
- Berger TW, et al. (2012) A hippocampal cognitive prosthesis: multi-input, multi-output nonlinear modeling and VLSI implementation. IEEE Trans Neural Syst Rehabil Eng 20(2):198-211.
- Song D, et al. (2018) Sparse Large-Scale Nonlinear Dynamical Modeling of Human Hippocampus for Memory Prostheses. IEEE Trans Neural Syst Rehabil Eng 26(2):272-280.
- Deadwyler SA, et al. (2013) Donor/recipient enhancement of memory in rat hippocampus. Front Syst Neurosci 7:120.
- De Ridder D, Elgoyhen AB, Romo R, & Langguth B (2011) Phantom percepts: tinnitus and pain as persisting aversive memory networks. Proc Natl Acad Sci U S A 108(20):8075-8080.
- Flesher SN, et al. (2016) Intracortical microstimulation of human somatosensory cortex. Science translational medicine 8(361):361ra141.
- Liu J, et al. (2015) Syringe-injectable electronics. Nat Nanotechnol 10(7):629-636.
- Zhou T, et al. (2017) Syringe-injectable mesh electronics integrate seamlessly with minimal chronic immune response in the brain. Proc Natl Acad Sci U S A 114(23):5894-5899.
- Schuhmann TG, Jr., et al. (2018) Syringe-injectable Mesh Electronics for Stable Chronic Rodent Electrophysiology. J Vis Exp (137).
- Neely RM, Piech DK, Santacruz SR, Maharbiz MM, & Carmena JM (2018) Recent advances in neural dust: towards a neural interface platform. Curr Opin Neurobiol 50:64-71.
- MacLean P (1990) The triune brain in evolution (Plenum Press, New York).