Demetrius Lopes has been working on the surface modification of stents and flow diverters, given that thromboembolic and antiplatelet-related complications represent the majority of those reported when using these devices. His aim is to decrease their thrombogenicity and mitigate the need for dual antiplatelet therapy. He outlines the current methods being used to lower thrombogenicity and theorises that future applications of surface modification will move beyond the realm of flow diverters.

The introduction of devices with surface modification represents a new chapter for the field of neurointervention. Device surface modification is the practice by which we can modify the physical, chemical or biological characteristics of the surface of a device. There are many reasons for creating coatings and making surface modifications to endovascular devices. Ultimately, each modification is trying to “enhance” or create a desired feature for safety or performance. Currently, the most widely accepted indications for device coatings and surface modifications are:
- Lower material thrombogenicity
– Electropolishing
– Silicon carbide
– Heparin
– Phosphorylcholine
- Bioabsorbable stents
- Corrosion resistance
– Passivation
– Electropolish
– Parylene coating
– Material choice, including stainless steel, cobalt-chromium, and nickel titanium.
- Increase lubricity
– Hydrophobic
– Hydrophilic
It is crucial we continue to work on improving profiles for device safety in the field of neurointervention. The introduction of flow diverters for the treatment of cerebral aneurysms shifted the focus from intrasaccular embolisation to luminal reconstruction. It took over two decades to bring the concept of flow diversion to clinical practice. The initial trials demonstrated feasibility and safety treating complex cerebral aneurysms such as giant and fusiform types.
Flow diversion also improved cerebral aneurysm long-term occlusion rates. Aneurysm recurrence used to be the Achilles heel of intrasaccular therapies, such as coils. Intraluminal devices generally require use of a dual antiplatelet regimen to reduce their thromboembolic risk. Thromboembolic and antiplatelet-related complications are amongst the most frequently reported when using stents and flow diverters. Thrombogenicity in endovascular intervention is a multifactorial issue. At least three main factors play a significant role in thrombus formation, also known as Virchow’s triad of thrombosis:
- Surface activation – Influenced by intimal damage and device material
- Flow Disruption – Determined by wall apposition and stent design
- Hypercoagulable state – Can come about due to a lack of response to antiplatelets or heparin-induced thrombocytopenia
Thus, the surface modification of flow diverters to decrease their thrombogenicity, and perhaps the need for dual antiplatelets, seems to be a natural next step to advance our field. Furthermore, as we move into the next generation of flow diverters, we will be faced with new challenges for expansion of indications for ruptured aneurysms and the more distal and smaller vessels. There are different methods to lower the thrombogenicity of a device. Some examples include:
- Electropolishing– EP nickel titanium absorb fewer platelets and plasma than stainless steel
- Silicon carbide – May reduce thrombogenicity
- Heparin coating – Actively prevents thrombus. May affect aneurysm occlusion
- Phosphorylcholine:
– Present in the surface of red blood cells. Device surface mimics red cell membrane
– Phosphorylcholine surface modification of the Pipeline Flex Embolisation Device (Medtronic; shield technology) was the first commercially available device for clinical use
Since there are multiple factors that determine the formation of thrombus in an endovascular implant, it is very challenging to simulate, in the bench, all variables using a single model. Therefore, it is worthwhile to review the possible types of bench testing these devices are being submitted to, prior to clinical trials.
Surface modified device performance test
- Delivery test
– Resistive forces are measured on the pusher as the implant is delivered through simulated vasculature.
- In vitro thrombogenicity assessment
– Thrombogram analysis
– Human blood loop
– Ex vivo primate shunt
- Animal models (porcine, rabbit) to evaluate thrombogenicity, endothelialisation, parent vessel stenosis, and aneurysm occlusion
– Histology (both acute and long-term)
– Optical Coherance Tomography (OCT)
We have had a very positive experience using OCT in a porcine model to follow the impact in the “healing” process of different surface modified devices. In our studies, the phosphorylcholine surface modification demonstrated a more evenly distributed concentric neointimal formation and no delay in endothelialisation compared to similar devices without phosphorylcholine surface modification.

over time in a porcine model. Day 0 (left) vs. day 14 (right).
At this point, the initial clinical experience with flow diverter with phosphorylcholine surface modification has demonstrated a good safety profile and no compromise on aneurysm occlusion rates (Table 1).
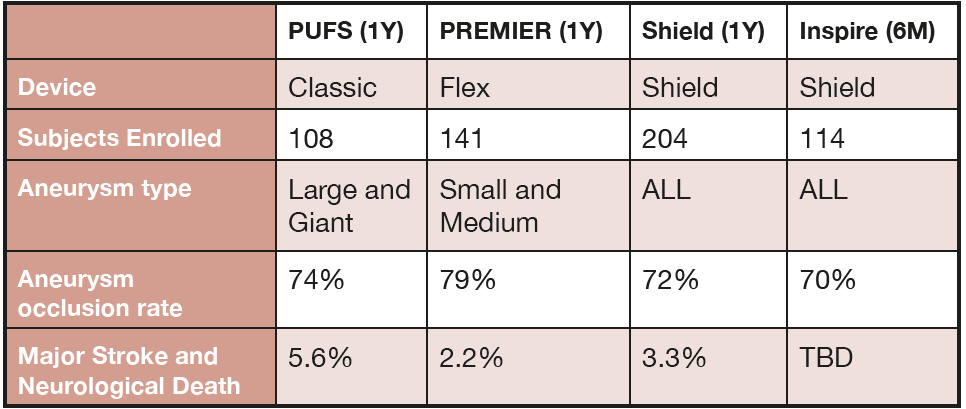
with surface modification (Shield and Inspire).
The preclinical testing of flow diverter surface modification has shown promising results. The translation of such results has been confirmed in initial clinical reports. It still remains to be seen if surface modification will decrease thromboembolic complications or antiplatelet medication needs compared to current devices.
The future possibilities with surface modification may also involve applying it to devices beyond flow diverters. We are already seeing new concepts with coatings and surface modification aimed towards decreasing thrombogenicity as well as faster endothelialisation.
Demetrius Lopes is a cerebrovascular neurosurgeon specialising in neuroendovascular therapy. At Advocate Health, he serves as medical director of the cerebrovascular and neuroendovascular programme, and is co-director of their stroke programme.









