Rapid Medical today announced US Food and Drug Administration (FDA) investigational device exemption (IDE) approval for what it claims is the first ever trial to expand interventional stroke treatment to distal regions of the brain.
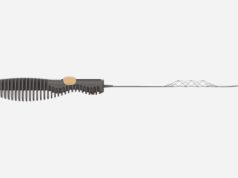
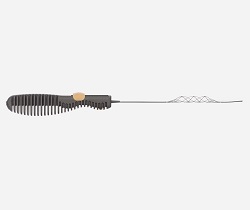
The DISTALS study is a pivotal, international, multicentre, randomised controlled trial (RCT) to evaluate the safety and effectiveness of distal thrombectomy. This new frontier for ischaemic stroke has been created by Rapid Medical’s development of TIGERTRIEVER 13—the smallest available thrombectomy device—according to a company press release.
“We are especially pleased to receive unprecedented FDA IDE approval for a study with DISTALS focus,” said Jeffrey Saver, professor of neurology and director of the Comprehensive Stroke and Vascular Neurology Program at the University of California Los Angeles (UCLA) in Los Angeles, USA, and the study’s principal investigator. “With the ever-growing benefits of mechanical thrombectomy, we hope to offer better recoveries to a much broader patient population. Rapid Medical has been at the forefront of enabling that progress.”
Currently, less than 10% of ischaemic stroke patients receive mechanical thrombectomy, a well-proven treatment that significantly improves patient outcomes, the release continues. While Rapid Medical’s TIGERTRIEVER 13 has already been used to treat nearly 1,500 patients in Europe and the Middle East, the DISTALS study aims to extend these benefits to an additional 25–40% of stroke patients. It focuses on far territories in the brain, such as M3 blood vessels, and allows intervention within 24 hours from symptoms onset.
“The DISTALS trial is critical for the field of interventional stroke therapy,” said David Fiorella, director of the Cerebrovascular Center at Stony Brook University Medical Center in Stony Brook, USA, and co-coordinating investigator. “It potentially allows us to offer thrombectomy to an additional large group of stroke patients who may benefit substantially.”
The leading investigators of the DISTALS study also include René Chapot, professor and department head of neuroradiology and radiology at Alfried Krupp Krankenhaus in Essen, Germany and Rishi Gupta, director of Neurocritical Care at Wellstar Health System in Marietta, USA. Rapid Medical plans to start the study in Q4 2021 or early in Q1 2022—according to the company’s co-founder and CEO Ronen Eckhouse.