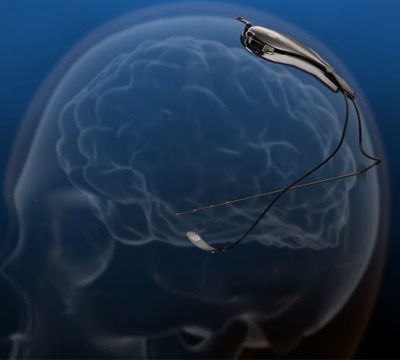
NeuroPace has closed a US$74 million round of funding led by the KCK Group and OrbiMed Advisors. This funding provides the resources necessary to rapidly expand commercialisation of the NeuroPace RNS System, an epilepsy device approved by FDA to treat adults with partial (focal) onset seizures that are not well controlled by medication alone.
The RNS System has been evaluated in multiple controlled, prospective studies. Outcomes are long-lasting and robust as evidenced by the 72% median seizure reduction patients experienced seven years after initiating the therapy, including 30% of patients who experienced seizure reductions of 90% or greater. Approximately 1,300 patients in the USA have been implanted with the system to date.
A company release explains that the RNS System is the first and only closed-loop brain-responsive neurostimulation system designed to prevent epileptic seizures at their source. The RNS System treats seizures by continuously monitoring brain waves, detecting unusual activity, and automatically responding with imperceptible electrical pulses before seizures occur. Physicians can programme the detection and stimulation parameters of the implanted RNS neurostimulator non-invasively to personalise therapy for each individual.
“With this additional funding, we now have the resources necessary to substantially accelerate adoption of RNS System and improve quality of life for hundreds of thousands of patients in the USA alone who live with uncontrolled, disabling seizures,” says Frank Fischer, NeuroPace CEO.