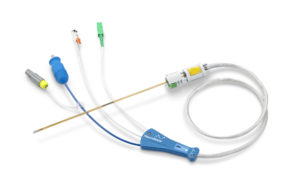
 Monteris Medical has announced it has received clearance from the US Food and Drug Administration (FDA) for its NeuroBlate Optic laser probe, a laser probe with fibre optic controlled cooling. The NeuroBlate Optic laser probe is for use with the NeuroBlate system, a minimally invasive MRI-guided robotic laser thermotherapy for use with brain tumours and epileptic foci.
Monteris Medical has announced it has received clearance from the US Food and Drug Administration (FDA) for its NeuroBlate Optic laser probe, a laser probe with fibre optic controlled cooling. The NeuroBlate Optic laser probe is for use with the NeuroBlate system, a minimally invasive MRI-guided robotic laser thermotherapy for use with brain tumours and epileptic foci.
Martin J Emerson, president and chief executive officer of Monteris Medical, said, “The Optic laser probe’s fibre optic temperature sensor retains the NeuroBlate system’s unique ability to deliver ablation and cooling control while also raising the probe’s safety profile.”
The NeuroBlate Optic laser probe platform was designed with patient safety in mind. The new design replaces the metal thermocouple inside the laser probe with a non-metallic fibre optic temperature sensor, thus eliminating any risk of unintended probe heating. MR scan restrictions issued as part of the field advisory notice in 2017 have been eliminated, as specific components of the NeuroBlate Optic laser probe in contact with the patient are non-metallic. Additionally, the non-metallic fibre optic temperature sensor allows for more freedom to customise the trajectory during surgical planning and positioning; a feature of which is especially useful for challenging target locations.
Today’s NeuroBlate system continues to feature a 1,064nm wavelength, which delivers controlled heating of targeted ablation zones, allowing for deeper penetration of energy; a slower rate of tissue heating, and the prevention of over-heated targeted tissue.
In addition to the new NeuroBlate Optic laser probe for the brain, the NeuroBlate system features exclusive TruTemp technology, which is built to deliver accurate thermography with enhanced visualisation. TruTemp technology uniquely mitigates the factors that may negatively influence MRI thermometry, providing confidence in the accuracy of the ablation zone and added safety assurance. NeuroBlate Fusion Software, the exclusive software intelligence behind the NeuroBlate system, delivers advanced image co-registration tools for assured surgical accuracy, enhanced visualisation of the ablation target, and maintains robotic control of the laser probe throughout the ablation.
Since receiving FDA 510(k) clearance in 2013, the NeuroBlate system has been used in more than 2,000 patient procedures across 60 installed systems in the USA and Canada, and continues to deliver precision and confidence, which are vitally important during an ablation procedure.
The NeuroBlate system is a minimally invasive robotically controlled laser thermotherapy that uses MRI-guided laser light to ablate unwanted tissue in the brain where the lesion, or abnormal tissue, originates. Unlike traditional brain surgery, a procedure with the NeuroBlate system does not require a large opening in the skull. Instead, doctors create a small hole in the skull, about the diameter of a pencil. While the patient is in the MRI machine, the doctor guides a small laser device (probe) through the hole into the lesion. The probe delivers laser light energy that heats and destroys the affected tissue. Because the NeuroBlate system is MRI-guided, the neurosurgeon is able to visualise the specific area of the brain to be ablated. The precise nature of the procedure helps to lessen the likelihood of harm to nearby healthy brain tissue.









