Magnus Medical has announced that the first participants have been treated in the Open Label Optimisation (OLO) clinical trial evaluating the effectiveness of the company’s neuromodulation system with SAINT technology for the treatment of major depressive disorder (MDD).
Magnus Medical has announced that the first participants have been treated in the Open Label Optimisation (OLO) clinical trial evaluating the effectiveness of the company’s neuromodulation system with SAINT technology for the treatment of major depressive disorder (MDD).
“SAINT therapy is the beginning of a revolution in psychiatry: fast, effective and personalised treatments that give hope to patients who have failed to receive relief from traditional approaches,” said David Carreon (Acacia Mental Health, Sunnyvale, USA), a primary investigator and the first to enrol in the OLO study at his centre. “It could profoundly change the way we care for those with treatment-resistant depression and be a model for addressing other conditions as well. Our clinic specialises in caring for the hardest and most hopeless, depressed patients by providing the best treatments available. We have started to enrol participants and are eager to find out whether the overwhelmingly positive results seen at the initial trials at Stanford will translate to our own patients.”
Up to nine clinics nationwide will participate in the OLO study, enrolling up to 1,000 adults suffering from a major depressive episode who have failed to receive satisfactory improvement from prior antidepressant medication in the current episode. Treatment will be delivered on an accelerated timeline—10 sessions a day, composed of 10-minute treatments of SAINT therapy with 50-minute breaks, for five consecutive days. Trial results are expected in 2027, as per a Magnus press release.
“We are delighted to have our first participants enrolled in the OLO study and happy to be working with Magnus to help provide a rapidly acting solution that may solve a fundamental problem in treating pervasive major depression,” said Robert Bota (Brain Health Solutions, Costa Mesa, USA), a primary investigator in the OLO study.
“We are very pleased to announce the start of the OLO clinical trial with SAINT therapy for people with MDD who are ineffectively treated by prior antidepressant medications,” added Brandon Bentzley, co-founder and chief scientific officer of Magnus. “Results from the previous randomised controlled trial were extremely promising—79% of participants experienced relief from their severe depression after receiving SAINT treatment. Given the enormous need for acute care in depression, we knew we needed to make SAINT treatment more widely available while building a body of clinical data. The OLO trial is the first time the SAINT neuromodulation system will be studied at multiple sites, allowing a large sample size of adults suffering from clinical depression to access the rapid-acting, personalised treatment for their treatment-resistant major depression.”
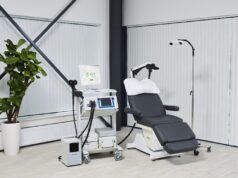
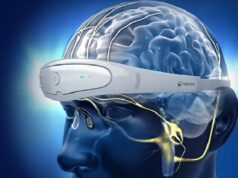
The release notes that the Magnus neuromodulation system with SAINT technology uses structural and functional magnetic resonance imaging (MRI) to inform a proprietary algorithm that identifies the optimal anatomic brain target for focused neurostimulation in people with MDD. This new approach is delivered on an accelerated five-day timeline and is “precisely tailored” to each person’s brain connectivity, whereas conventional treatments for this patient sector take weeks or even months to ease symptoms, Magnus also claims.