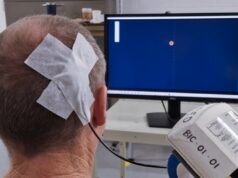
Cognito Therapeutics has announced the first patient enrolment in its US pivotal study, HOPE, which is designed to demonstrate the safety and efficacy of the company’s proprietary, non-invasive stimulation device—CogTx-001—in patients with Alzheimer’s disease.
Cognito Therapeutics has announced the first patient enrolment in its US pivotal study, HOPE, which is designed to demonstrate the safety and efficacy of the company’s proprietary, non-invasive stimulation device—CogTx-001—in patients with Alzheimer’s disease.
The HOPE study for Alzheimer’s disease is evaluating a novel wearable device developed by Cognito that has the potential to slow the progression of Alzheimer’s disease—without the safety concerns associated with medications or highly invasive procedures. In the blinded, sham-controlled study, 400 patients with Alzheimer’s will use the company’s non-invasive wearable device at home, on a daily basis, for 12 months.
Cognito’s device has been assessed in multiple clinical studies for safely and effectively delivering specific frequencies of targeted light- and sound-based stimulation, as per a press release. In the OVERTURE study, patients using it reported an 83% reduction in disease progression, as measured by the Mini-Mental State Exam (MMSE), and 78%, as measured by the Alzheimer’s Disease Cooperative Study-Activities of Daily Living (ADCS-ADL). A significant reduction of brain atrophy and preservation of white matter was also reported in patients using the Cognito device over a six-month period.
The 12-month HOPE study is designed to build upon the success of the OVERTURE study by demonstrating both slowing of disease progression—as measured by the primary endpoints of the ADCS-ADL, and a combined statistical test (CST) of the ADCS-ADL and the MMSE—as well as slowing of brain atrophy on magnetic resonance imaging (MRI). The study is also designed to include assessments of secondary endpoints including change from baseline in the Clinical Dementia Rating-Sum of Boxes (CDR-SB).
“The HOPE study offers a rare opportunity for patients with mild-to-moderate Alzheimer’s disease to participate in a research study utilising technology as an intervention,” said Michelle Papka (Cognitive and Research Center of New Jersey, Springfield, USA). “Patients and their family members have expressed great interest in enrolling in this study because it is non-invasive, and represents a novel and safe approach to potentially slowing the progression of Alzheimer’s disease.”
Cognito’s device is limited by US law to investigational use only and is not available for sale, the company release adds.