 Defining true success as “being able to decrease or even eliminate a patient’s dependency on oral opioids”, Corey W Hunter, founder of Ainsworth Institute of Pain Management (New York, USA) discusses the rationale and achievements of Burst-DR spinal cord stimulation (SCS), as he ultimately deduces that “a failure in SCS is not necessarily a failure of the therapy as a whole”, but “merely a failure of one particular type of stimulation”.
Defining true success as “being able to decrease or even eliminate a patient’s dependency on oral opioids”, Corey W Hunter, founder of Ainsworth Institute of Pain Management (New York, USA) discusses the rationale and achievements of Burst-DR spinal cord stimulation (SCS), as he ultimately deduces that “a failure in SCS is not necessarily a failure of the therapy as a whole”, but “merely a failure of one particular type of stimulation”.
SCS has been the “go-to” therapy for interventional pain management doctors to treat patients with intractable, chronic pain for decades. While providing pain relief and improvements in function, perhaps its most valuable attribute is its ability to serve as an alternative for opioids. Despite the fact that SCS is over 50-years-old, the bulk of the innovation surrounding this therapy has really only taken place in the last 10-15 years. In previous years, SCS was more of a one-size-fits-all therapy with little-to-no variability or customisability for the individual or diagnosis. Because of that, not every patient was a success. Patients would fail their SCS devices over time and be left with no other options than either getting an intrathecal pump implanted or simply relegating themselves to a life of oral opioids.
At present, there are a number of distinctly different variations within SCS that give the user more “options” to choose from for a given patient. These options have translated into increased overall success rates for SCS as a whole, as well as overall pain relief.
Despite these vast improvements, there are still those patients that may fail SCS and want to know what their options are. The intuitive answer is if a patient fails SCS-“option A” to switch the person to SCS-“option B,” however there is no evidence to validate this theory. In cardiology, if a patient with hypertension fails with beta-blockers, the next step is to try a different class of medications, perhaps a calcium-channel blocker—not give up on the patient and accept that the hypertension is untreatable. With the current advances in SCS, the there are significant enough differences between the options that one could make the argument that they are different therapies and would fall in line with the same rationale as switching a patient from a beta-blocker to a calcium-channel blocker.
In 2016, St. Jude Medical (now known as Abbott) introduced a new waveform for SCS known as Burst-DR. This innovation was based on the idea that some neurons communicate using different electoral signals that travel in packets or “bursts” which will translate into a different type of action potential being propagated; in an effort to capture these specific neurons and provide more complete pain relief, burst-capable systems will mimic that electrical signature and cause those neurons to fire as well. On an physiologic level, this is a significant departure from conventional SCS systems and sparked the conversion as to whether or not one could consider SCS with burst to be a different enough therapy that it would be akin to trying a different class of medications as described in the example above for a patient with hypertension. In other words, failing one type of spinal cord stimulation is not necessarily a failure of the therapy as a whole, just that one particular “class.”
The BURST(able) study was a physician-initiated research initiative that specifically evaluated if the burst waveform as defined by Dirk DeRidder could be used in the setting of SCS failure to “salvage” the patient and bring them back to a point where they were reporting pain relief again. In addition to pain relief, we also evaluated the effect this specific waveform had on opioid usage—as described above, when a patient begins to find themselves no longer responding to their SCS system, they will turn or return to using opioids. While a patient reporting that their pain is “better” by changing from one SCS therapy to another would certainly be of interest to the field, the true accomplishment lies in being able to decrease or even eliminate a patient’s dependency on oral opioids.
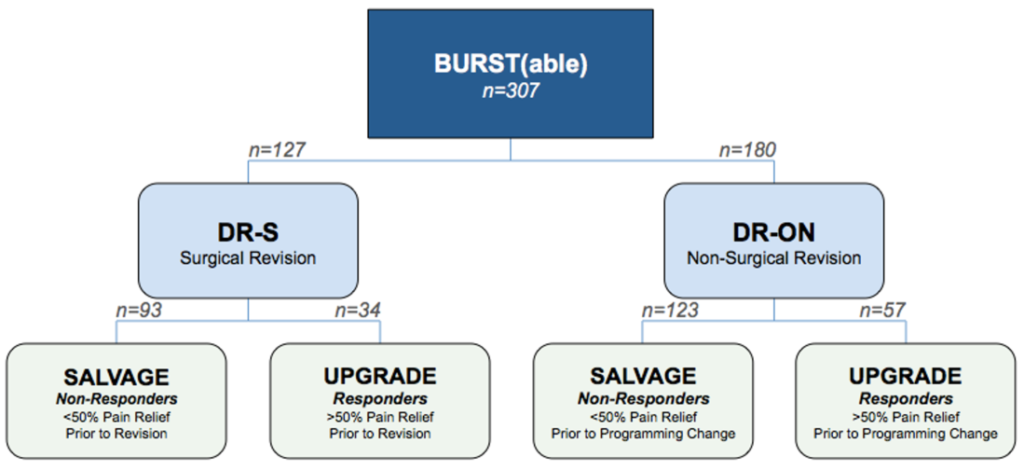
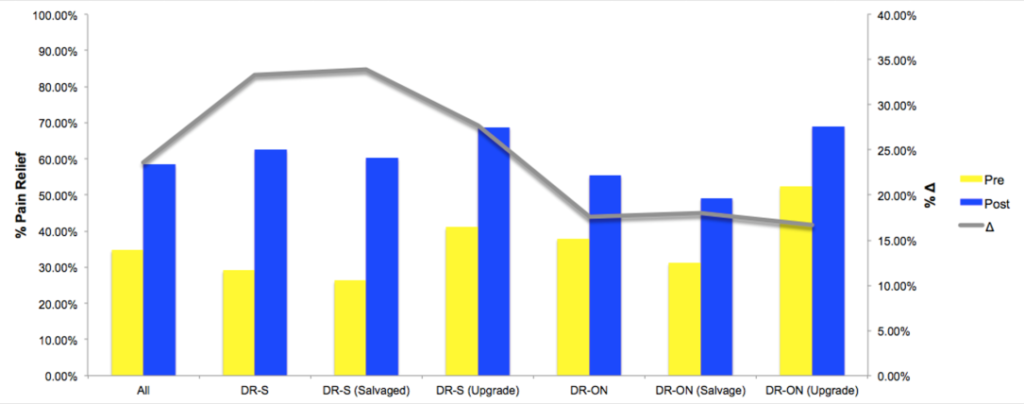
There were a total of 307 patient-entries across seven sites—all of which were switched from their current SCS platforms to one that was burst-capable (127 were surgically revised [DR-S] and 180 had their devices programmed [DR-ON]). There were statistically significant reductions in pain across the whole study, each of the main cohorts (DR-S vs. DR-ON) as well as the sub-cohorts (salvage vs. upgrade). In the surgically revised group (DR-S), some patients had their entire system changed out (leads and battery) where as others only had the battery swapped while using adaptors to hook into the old leads. The patients in the study have been followed out to nearly one-year and the statistically significant improvements in pain have been shown to hold up.
The study showed no significant differences in pain relief between the two groups, which is good news for patients as they can go with the less invasive option of simply getting a different battery, and expect the same degree of improvement. The most important improvement found in this study was the statistically significant reduction in opioid use across the entire cohort of patients. There is little satisfaction to be gleaned from reducing a patient’s pain if one cannot also decrease their opioid use. This study showed that not only were patients reporting improvements in pain simply by being exposed to the burst waveform, but they found themselves needing less opioids to manage what pain they had left.
The findings in this study help validate a number of theories as it pertains to neuromodulation. Firstly, a failure in SCS is not a failure of the therapy as a whole, merely a failure of one particular type of stimulation. Secondly, when presented with a patient who is failing SCS, changing to a different stimulation model like burst is a valid, evidence-based treatment option. And lastly, SCS can be a valid means to reduce opioid consumption in chronic pain patients.
While the results in the BURST(able) study are meaningful, a prospective study would be the ideal means to measure these theories. Nonetheless, it is an important first step and certainly creates a whole new conversation as it applies to the field, where neuromodulation should be placed in the treatment algorithm.

Corey W Hunter is the Executive Director and founder of the Ainsworth Institute of Pain Management (New York, USA).










