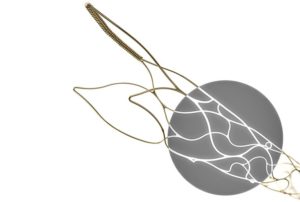
phenox has announced the full European launch of the pRESET 5–40 and pRESET LUX 4–20 thrombectomy devices after receiving CE mark approval last year. The pRESET device is a stentriever which is used for mechanical thrombectomy in patients with acute ischaemic stroke, due to large vessel occlusion.
Over the past few years, safety and efficacy using mechanical thrombectomy has been proven in patients with large vessel occlusion in four major randomised controlled trials (MR CLEAN1, SWIFT-PRIME2, EXTEND-IA3, ESCAPE4). The pRESET thrombectomy device was evaluated in the ARTESp study (prospective, multicentre, single-arm post-market clinical follow-up study) and demonstrated its safety and efficacy: TICI 2b/3: 84,4% and mRS (0–2) at 90 days: 62.5%.
According to the company, the development of the new pRESET 5–40, addresses the trend towards the use of longer thrombectomy devices, which may have the capability to remove an even larger clot mass in fewer attempts. The 5–40 product line extension inherits the principal pRESET design, including a low-profile 0.021” delivery platform.
In addition to the 5–40 line extension, the pRESET product family is broadened with the new pRESET LUX technology, which adds full radiopacity to the proven stentriever design.
A recent news release also states that a noble alloy coating (NAC) has been applied to the full length of the device, so that it can be fully seen under X-ray. By maximising the device’s visibility, the exact positioning and opening can be evaluated by the operating physician.










