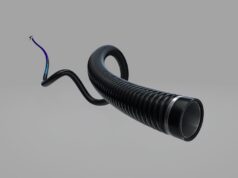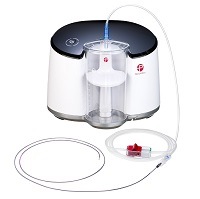
Penumbra has announced US Food and Drug Administration (FDA) 510(k) clearance and commercial availability of the RED 62 reperfusion catheter—the latest addition to the company’s comprehensive Penumbra system. RED 62 is designed to navigate complex distal vessel anatomy and deliver powerful aspiration, alongside the Penumbra ENGINE, for the removal of blood clots in acute ischaemic stroke patients with large vessel occlusions.
“In my initial experience with RED 62, I am impressed by how responsive the catheter is as I navigate through the intricate vessels of the brain,” said Kurt Reuland, interventional neuroradiologist at CHRISTUS Trinity Mother Frances Health System (Tyler, USA). “The performance and length of RED 62 enable me to address a broader group of patients using aspiration thrombectomy to remove blood clots, and improve my patients’ health outcomes.”
The RED 62 is engineered with the latest innovations in tracking and aspiration technology to address large vessel occlusions located in more challenging distal vessel anatomies while maximising powerful aspiration to remove blood clots, according to a Penumbra press release. It has a low diameter profile and extended length to reach the target vessel.
“With the continued success of aspiration thrombectomy, physicians asked us to address navigation in more challenging distal vessels,” said Adam Elsesser, president and CEO of Penumbra. “RED 62 builds on our previous design generations, continuing our efforts to maximise both trackability and aspiration power. This is the first catheter in the RED series, which will provide physicians with the broadest portfolio of aspiration thrombectomy solutions for stroke management while also focusing on reducing overall healthcare costs.”