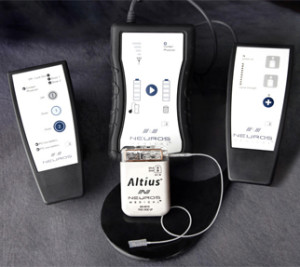
Neuros Medical has announced that it has successfully completed a planned interim analysis of its pivotal study. The study is designed to evaluate the Altius System High Frequency Nerve Block technology for the management of post-amputation pain.
As part of the interim analysis, an independent data monitoring committee conducted a statistical probability analysis focused on safety as well as efficacy factors for the first 20 patients enrolled. Based on the results, the committee recommended continuation of the study.
“The interim analysis provides hope for this promising therapy and for a successful pivotal study. The Neuros High Frequency Nerve Block therapy is truly a paradigm shift in treating chronic pain, especially post-amputation pain, due to its on-demand blocking effect,” says Leonardo Kapural, the study’s principal investigator, from the Carolinas Pain Institute in Winston-Salem, USA.
“The successful completion of the interim analysis marks an important step in our goal of providing a proven, safe, and effective therapy for patients suffering from chronic pain,” states Jon J Snyder, president and chief executive officer of Neuros Medical, “And is a testament to the commitment of our pivotal study principal investigators and clinical research coordinators, as well as the team at Neuros.”
The prospective, randomised, controlled pivotal clinical trial will consist of up to 130 patients at 15 institutions to evaluate the safety and efficacy of Neuros Medical’s Altius System. When completed, the results will support a PreMarket Approval Application to the FDA in order to market the device. The pivotal study builds off of Neuros’ long-term pilot study, in which patients are nearing four years of device use and over 4,300 uses to date, and patients continue to report significant pain reduction. In addition, more than half of the responders discontinued their pain medication use during the pilot study.









