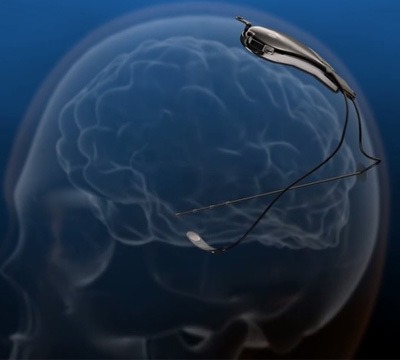
NeuroPace has announced that its closed-loop responsive neurostimulation (RNS) system has won the ‘Most Promising New Product’ award at the 2015 Phoenix Medical Device CEO Conference.
The RNS system is composed of the implantable RNS neurostimulator, and two leads which should be placed near seizure onset areas. The closed-loop RNS system monitors the brain and provides stimulation when needed, unlike other existing therapies which stimulate the brain without assessing the need for treatment and analysing the brain’s response.
“We are honoured that our RNS System was recognised by industry leaders at the 2015 Phoenix Medical Device CEO Conference,” says Frank Fischer, CEO of NeuroPace. “Beyond epilepsy, we believe there is immense potential for the RNS System to be used in the treatment of other neurological disorders.”
According to NeuroPace, the RNS system also provides physicians with clinically meaningful ongoing data about their patients’ seizure frequency and brain electrical activity. Each patient has a remote monitor at home to wirelessly collect and upload data from their neurostimulator. The data is made available to their doctor to review and analyse to help improve patient care. NeuroPace claims that for many medically refractory partial-onset epilepsy patients who are not candidates for epilepsy surgery, the RNS system provides a clinically proven therapy option to reduce seizure frequency and improve quality of life.
Long-term benefits of the RNS System were recently published in Neurology, demonstrating that the it can significantly reduce seizure frequency among adults who have a common form of epilepsy that is difficult to treat with medication. The published interim study results include data on 230 people with medically intractable partial onset epilepsy enrolled at 33 Comprehensive Epilepsy Centers in the United States. The study confirmed the long term effectiveness of RNS System therapy. Median seizure frequency reductions compared to patients’ pre-implant seizure frequencies ranged from 60-66% in years three through six post-implant. At this time, some patients have been treated with the RNS system for more than 11 years, and more than 1,700 patient years of experience with responsive neurostimulation have been accumulated to date.
Findings from another study, recently published in Epilepsia, demonstrated that people treated with the RNS System showed no adverse effects on cognition.
The RNS system was FDA approved in November 2013 to treat adults with partial onset seizures that are not controlled by medication.













