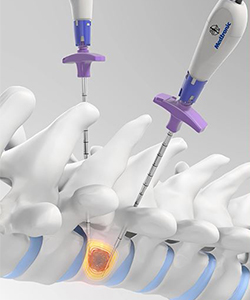
Medtronic is to launch the OsteoCool radiofrequency ablation system for surgeons who treat patients with painful spinal metastases. The system has been granted US Food and Drug Administration (FDA) 510(k) marketing clearance.
According to a Medtronic press release, the Osteocool system is the only cooled radiofrequency (RF) ablation technology that offers concurrent dual-probe capabilities. This is intended to offer procedural flexibility, and predictable, customisable treatment.
“The most common complaint associated with spine metastases is pain – it’s often progressive and significantly reduces the patient’s quality of life,” says Sandeep Bagla, an interventional radiologist with the Vascular Institute of Virginia in Woodbridge, USA. “With the OsteoCool system, I can now ablate the patient’s painful tumour with a reproducible procedure that’s appropriate for their unique anatomy.”
Julie Foster, general manager and vice president of the Pain Therapies business in the Neuromodulation division, which is part of the Restorative Therapies Group at Medtronic, says, “Our customers treat patients with Kyphon balloon kyphoplasty who also have painful spinal metastases. The OsteoCool system gives physicians a way to treat that pain in a single procedure with a familiar, minimally invasive technique.”
In November, Medtronic received expanded indications for Kyphon Xpede bone cement, which now includes clearance for the treatment osteolytic lesions. Where indicated, the OsteoCool bone access kit can be used for a subsequent, physician-directed procedure such as cementoplasty.
Medtronic acquired the OsteoCool technology and associated intellectual property from Baylis Medical on December 16, 2015 and partnered with the company to further innovate the system. Terms of the acquisition were not disclosed.
The system is temperature controlled and uses internally water-cooled probes to prevent overheating of surrounding tissue during the procedure. The 17-gauge, bipolar probes are available in three lengths and may be used through a variety of cannula sizes. Because two OsteoCool RF ablation probes can be used simultaneously, the system should support a variety of ablation scenarios accommodating unique patient and procedural needs. The device is currently pending CE marking.










