The US Food and Drug Administration (FDA) has authorised marketing of a new device indicated for use in patients aged 18 and older undergoing stroke rehabilitation to facilitate muscle re-education, and for maintaining or increasing range of motion. The IpsiHand Upper Extremity Rehabilitation System (Neurolutions) is a brain-computer interface (BCI) device that assists in rehabilitation for stroke patients with upper extremity—or hand, wrist and arm—disability.
The US Food and Drug Administration (FDA) has authorised marketing of a new device indicated for use in patients aged 18 and older undergoing stroke rehabilitation to facilitate muscle re-education, and for maintaining or increasing range of motion. The IpsiHand Upper Extremity Rehabilitation System (Neurolutions) is a brain-computer interface (BCI) device that assists in rehabilitation for stroke patients with upper extremity—or hand, wrist and arm—disability.
“Thousands of stroke survivors require rehabilitation each year,” said Christopher M Loftus, acting director of the Office of Neurological and Physical Medicine Devices in the FDA’s Center for Devices and Radiological Health. “Today’s authorisation offers certain chronic stroke patients undergoing stroke rehabilitation an additional treatment option to help them move their hands and arms again, and fills an unmet need for patients who may not have access to home-based stroke rehabilitation technologies.”
According to the American Centers for Disease Control and Prevention (CDC), someone in the USA has a stroke every 40 seconds, and about 795,000 people in the USA have a stroke each year. Post-stroke rehabilitation, however, helps many of these individuals overcome disabilities that result from stroke damage.
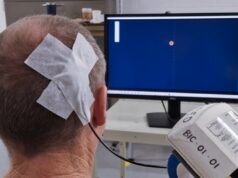
The IpsiHand System uses non-invasive electroencephalography (EEG) electrodes, instead of an implanted electrode or other invasive feature, to record brain activity. The EEG data is then wirelessly conveyed to a tablet for analysis of the intended muscle movement (intended motor function) and a signal is sent to a wireless electronic hand brace—which in turn moves the patient’s hand. The device aims to help stroke patients improve grasping. It is currently available as a prescription-only option making up part of a patient’s rehabilitation therapy.
The FDA stated in a press release that, having previously granted Breakthrough Device designation, it assessed the safety and effectiveness of the IpsiHand System through clinical data submitted by Neurolutions, including an unblinded study of 40 patients over a 12-week trial. All participants demonstrated motor function improvement with the device throughout the trial, while adverse events reported included minor fatigue and discomfort, and temporary skin redness.
The US regulator also noted the IpsiHand System device should not be used by patients with severe spasticity or rigid contractures in the wrist or fingers that would prevent the electronic hand brace from being properly fitted or positioned for use, or by those with skull defects due to craniotomy or craniectomy.