MicroVention, a US based subsidiary of Terumo and a global neurovascular company announced the US FDA premarket approval (PMA) for the FRED (Flow re-direction endoluminal device) device for the treatment of brain aneurysms.
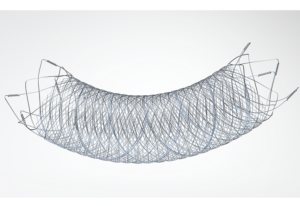
The FRED device uses a self-expanding braided nitinol mesh to help re-direct blood flow and promote aneurysm occlusion. The interwoven nitinol design of the FRED device allows for smooth delivery to the target aneurysm, as well as reliable opening and vessel wall apposition, resulting in high treatment durability.
“The FRED device represents a step forward in flow diversion technology with the inclusion of the lowest profile delivery platform in the USA. This advancement will allow physicians to more easily access and treat wide-necked or fusiform aneurysms,” said Cameron McDougall, director of endovascular neurosurgery and professor of neurosurgery at The Johns Hopkins Hospital (Baltimore, USA), and primary investigator for the FRED pivotal trial. “The pivotal study shows that a single FRED device is safe and effective for use in a variety of aneurysm sizes and locations and expands treatment options for patients.”
The FRED device has been CE marked since 2013, safely used in nearly 20,000 procedures, and published in numerous clinical studies around the world. The FRED device pivotal study adds new evidence to the large existing body of global clinical data, further demonstrating the safety and effectiveness of the device.
“The FRED device helps address a need for a clinically proven flow diverter with simplified delivery,” said Irina Kulinets, vice president of World Wide Regulatory Affairs, Clinical Research & Quality at MicroVention.










