A study involving virtual participants—rather than real patients—was “as effective as traditional clinical trials” in evaluating the treatment of brain aneurysms using a flow diverter, according to new research. These findings, which are published in the journal Nature Communications, have been described as a proof of concept for “in-silico trials” where, instead of recruiting people to a real-life clinical trial, researchers build digital simulations of patient groups.
In-silico trials could “revolutionise the way clinical trials are conducted”, a press release from the University of Leeds in the UK states, by reducing the time and costs of getting new medical devices and medicines developed, while also reducing human and animal harm in testing.
The virtual patient populations are developed from clinical databases to reflect age, sex and ethnicity, but they also simulate the way disease affects the human body—for example, the interactions between anatomy, physics, physiology, and blood biochemistry—before those simulations are then used to model the impact of therapies and interventions.
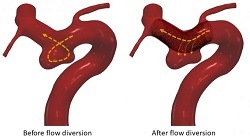
This international research, led by the University of Leeds, investigated whether an in-silico trial could replicate the results of three real-life clinical trials that assessed the effectiveness of a flow diverter device, which is a small, flexible mesh tube used to treat brain aneurysms by redirecting blood and reducing flow into the aneurysm—initiating a clotting process that eventually cuts the aneurysm off from blood circulation, thus healing it.
To establish their proof of concept, the university researchers had to see if the results from their in-silico study agreed with the results from three previous major clinical trials into the effectiveness of flow diverters.
Participants in the virtual trial
The researchers built a virtual population using real patient data drawn from clinical databases, ensuring that the anonymised virtual patients closely resembled the patients used in real flow diverter clinical trials in terms of age, sex and aneurysm characteristics.
The researchers then built a computational model that analysed how the implanted device would affect blood flow in each of the virtual patients. They were able to study different physiological conditions for each patient, such as normal and high blood pressure, and perform analyses on patient sub-groups, such as those with large aneurysms or aneurysms with a branch vessel.
The in-silico trial had 82 virtual cases, while the selected traditional clinical trials (PUFS [Pipeline for uncoilable or failed aneurysms], PREMIER [Prospective study on embolisation of intracranial aneurysms with the pipeline device], and ASPIRe [Aneurysm study of pipeline in an observational registry]) featured 109, 141 and 207 patients, respectively. Around half the cases in the traditional trials had high blood pressure.
The results of the in-silico trial predicted that 82.9% of the virtual patients with normal blood pressure would be successfully treated with a flow diverter. In the traditional clinical trials, the number of people who were successfully treated was 86.8%, 74.8% and 76.8%, respectively, thus showing that the in-silico trial replicated the traditional clinical trials’ results, according to the University of Leeds release.
In-silico trials hold “huge potential”
Alex Frangi, Diamond Jubilee Chair in Computational Medicine and Royal Academy of Engineering Chair in Emerging Technologies at the University of Leeds, who supervised the study, said: “The results demonstrate the huge potential of in-silico trials. We have shown that the approach can replicate the findings of traditional clinical trials—and they do that in a fraction of the time it normally takes, and at a fraction of the cost.”
In the in-silico trial, the researchers were also able to adapt their computational model to investigate new hunches or emerging scientific findings. And, by modelling the physics of blood flow and the biochemistry of blood clotting in aneurysms under different physiological conditions, they were able to identify sub-groups of patients at higher risk of stroke, with patients with large, complex-shaped aneurysms at higher risk of haemorrhagic stroke and patients with hypertension at higher risk of ischaemic stroke.
Being able to rigorously analyse medical device performance in this way will allow biomedical engineers to optimise the device design and reduce the risks associated with treatment, the release adds.
“In-silico trials offer an opportunity to do virtual experiments that could explain concepts that are difficult to study in conventional clinical trials,” Frangi added. “The current approach to improve our understanding of new medical devices is slow, as conventional trials can easily take five-to-eight years, from their design to completion. In-silico trials could reduce this period to less than six months in some circumstances, making knowledge and therapeutic technologies safer, and more promptly available to clinicians and patients.”
This research involved an international collaboration of scientists from Leeds, the University of Oxford (Oxford, UK), Radboud University Medical Centre (Nijmegen, The Netherlands), and KU Leuven (Leuven, Belgium). It was funded through the Royal Academy of Engineering (INSILEX), @neurIST from the European Commission, and the Engineering and Physical Sciences Research Council Centre for Doctoral Training on Fluid Dynamics.









