Codman Neuro has announced the acquisition of Pulsar Vascular Inc, a privately-held company with breakthrough platform technology for the neurovascular treatment of complex aneurysms. DePuy Synthes Products Inc, an affiliate of Codman Neuro, is the acquiring entity.
A Codman Neuro press release states that “this acquisition marks the latest expansion of its neurovascular portfolio and will complement a comprehensive portfolio of products for haemorrhagic and ischaemic stroke.” Financial terms of the transaction were not disclosed.
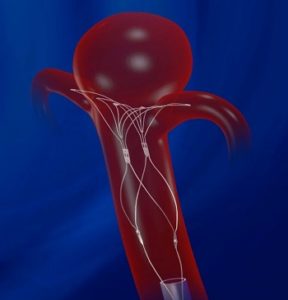
Founded by Sanderling Ventures in 2005, Pulsar Vascular Inc developed the PulseRider minimally invasive, self-expanding nitinol implant that is used to bridge the neck of cerebral aneurysms during treatment of unruptured wide-neck intracranial aneurysms originating on or near a bifurcation. A unique patented “saddle” shape stent leaves minimal metal in the parent artery, enabling easy access to the aneurysm for coiling while maintaining support.
According to the Brain Aneurysm Foundation, cerebral aneurysms claim nearly 500,000 lives worldwide each year, with half of the victims younger than 50. Many of these aneurysms develop in vascular bifurcations where the arteries branch, which may be difficult to treat with conventional devices.
“There is a significant unmet medical need for the treatment of complex bifurcation aneurysms which pose a high risk to the patient,” said Shlomi Nachman, company group chairman of Johnson & Johnson Medical Devices Cardiovascular & Specialty Solutions. “Pulsar Vascular Inc’s technology design is a great example of how innovation is driving improved outcomes for patients facing a condition that could have profound impact on their lives. Working together, we will leverage our proven market expertise and strong commercial footprint to bring this technology to many more clinicians worldwide.”
The PulseRider device received CE mark certification in Europe in 2013 and is currently sold in nine countries. Codman Neuro was previously the exclusive distributor of Pulsar Vascular Inc’s technology in Europe, Middle East and Africa. Pulsar Vascular Inc has completed a US IDE clinical trial and awaits US HDE approval.









