During a presentation at the European Society of Minimally Invasive Neurological Therapy annual meeting (ESMINT; 10–12 September, Nice, France) Jens Fiehler (director of the Department of Diagnostic and Interventional Neuroradiology, University Medical Center Hamburg-Eppendorf, Hamburg, Germany) said that with the changes made to visibility and vessel wall apposition from Enterprise to Enterprise 2 (Codman Neuro), the problems with the older generation device have been solved.
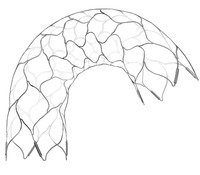
The Enterprise 2 vascular reconstruction device is designed to improve vessel wall conformability in tortuous anatomy, while maintaining its ability to provide a stable structure at the neck of an aneurysm, securing the placement of coils and maintaining blood flow through the artery.
In terms of what is required from a regular stent for aneurysm coiling support, Fiehler maintained that the needs have not evolved that much over the years. Those needs are: deliverability and manoeuvrability, retrieveability and repositioning capabilities, and visibility. Additionally, there is a need for good metal surface coverage allowing for a good scaffold for good coil support, and a stent should also have some flow diverting capabilities. Finally, he said, a stent should have good vessel wall apposition to avoid later thromboembolic events and migration of the stent.
He added that while the first generation Enterprise device is still a very good device, there were two areas where there was room for improvement—visibility and vessel wall apposition. This is largely where the improvements in the Enterprise 2 lie, he explained.
“In terms of visibility, the tantalum markers in Enterprise that were not so good for visibility, have been replaced in Enterprise 2 by platinum markers and the effect is really convincing. Additionally, changes to the variable strut width, increased expansion diameter (5mm) and amplitude of the sine wave have some implications for the behaviour of the device in the curve and works toward better applicability and better wall apposition.
“If we look at the two problems that Enterprise used to have—visibility and vessel wall apposition—I think both of them are solved; and the question of whether this is an evolution: I think it is intelligent design,” Fiehler concluded.
The Enterprise 2 vascular reconstruction device will now be studied in Europe in the SUNRISE2 study. SUNRISE2 is a prospective, multicentre, single arm, post-market study to confirm the clinical performance and safety of the Enterprise 2. There will be a minimum of 50 patients enrolled, 85% are expected to have non-ruptured aneurysms. The study will take place at up to 12 centres in Europe. The primary endpoints are absence of neurological complications and successful aneurysm treatment without retreatment, evaluated at six months.