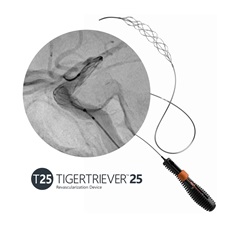
Rapid Medical today announced the US Food and Drug Administration (FDA) clearance of its Tigertriever 25 device—the latest addition to its Tigertriever platform—along with the first clinical cases demonstrating its performance in treating large vessel occlusion (LVO) stroke.
Designed specifically for large clot burden in proximal vessels, Tigertriever 25 combines a 53mm capture length with proprietary real-time force control. Rapid states in a press release that this capability, which is unique to the Tigertriever platform, enables physicians to actively control clot interaction during procedures, driving consistent performance across a wide range of clinical scenarios—including long thrombi and stenotic vessels—where conventional devices are “limited”.
“With every thrombectomy, we aim at our first pass being the last,” said Edgar Samaniego (University of Iowa, Iowa City, USA), who performed the first US case. “The Tigertriever 25 demonstrated excellent conformability to the vessel anatomy, accommodating the MCA [middle cerebral artery] diameter with a smooth transition into the terminal ICA [internal carotid artery]. This adaptability allows for effective engagement across long segments and makes it particularly valuable in LVOs with high clot burden—even in challenging, angulated anatomy.”
Rapid says Tigertriever 25 is “the largest thrombectomy device available”, having been specifically engineered to address occlusions in proximal vessels like the ICA and the M1 segment of the MCA, which are considered to be among the most challenging cases in stroke intervention. The device combines extended capture length, active operator-controlled expansion, and a distal net designed to maintain clot engagement over long segments, all of which are “key factors” in reducing clot fragmentation and distal embolisation, according to the company.
“Acute ischaemic stroke intervention today demands a diverse and adaptable toolkit to effectively address the wide spectrum of anatomical and clinical scenarios we encounter, and I am very impressed with the versatility of the Tigertriever 25 device,” commented Demetrius Lopes (Advocate Health Care, Chicago, USA). “Its design streamlines the procedural approach and has the potential to simplify the pathway to achieving first-pass success—an outcome that remains critical for improving patient outcomes in mechanical thrombectomy.”