Axonics has disclosed positive top-line results from the ARTISAN-SNM pivotal study, designed to gain marketing approval from the US Food and Drug Administration (FDA) for Axonics’ implantable sacral neuromodulation (SNM) devices for the treatment of urinary and bowel dysfunction.
The clinical study demonstrated that patients implanted with the Axonics r-SNM System received clinically meaningful and statistically significant improvements in urinary urgency incontinence symptoms and quality of life. Additionally, the study met all secondary endpoints. No serious device-related adverse events have been reported.
The ARTISAN-SNM study is a 129-patient single-arm, prospective, multicentre, unblinded pivotal clinical study approved under an FDA Investigational Device Exemption to evaluate the safety and efficacy of the Axonics r-SNM System for urinary dysfunction. The study was conducted in 14 centres in the US and five centres in Western Europe.
Karen Noblett, Chief Medical Officer of Axonics, commented, “Historically, SNM therapy has only been available with a non-rechargeable implanted neurostimulator with an average lifespan of four years requiring replacement surgery due to depletion of the neurostimulator battery. We believe that, in addition to reducing costs for patients and payers, the miniaturised 5cc rechargeable Axonics system, qualified to last in the body for at least 15 years, can significantly increase adoption of SNM therapy.”
Top-line results
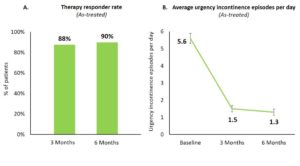
All patients diagnosed with urinary urgency incontinence and meeting study criteria were implanted with a tined lead and the Axonics neurostimulator. Efficacy data was collected using a three-day bladder diary, a validated quality of life questionnaire (ICIQ-OABqol), and a satisfaction questionnaire. Therapy responders were identified as patients with at least 50% reduction in urgency incontinence episodes at follow up visits as compared to baseline. An as-treated analysis was performed for all 129 implanted patients.
At six months, 90% of all implanted patients were therapy responders (Figure 1A) including 80% of therapy responders with a ≥75% reduction in urgency incontinence episodes of which 34% were completely dry.
Across all patients, urgency incontinence episodes per day reduced from 5.6±0.3 (mean ± standard error) at baseline to 1.3±0.2 at six months (p<0.0001; Figure 1B).
Patients averaged statistically and clinically significant improvement on the composite ICIQ-OABqol score (34 points) at six months as compared to baseline and 93% of all implanted patients were satisfied with their r-SNM therapy.
Axonics filed a PMA application on 3 December, 2018 and interim clinical data from the ARTISAN-SNM study with the FDA at the end of 2018. The clinical data disclosed herein has not yet been reviewed by the FDA. Axonics intends to file the six-month clinical data with the FDA during the week of 18 February. Axonics does not anticipate that the filing of this additional clinical data will impact the standard 180-day PMA review timeline for the FDA to complete its review and issue a decision letter.