 Methodist Le Bonheur Healthcare (MLH) is the first hospital system globally to participate in a landmark study using a first-of-its-kind EEG device.
Methodist Le Bonheur Healthcare (MLH) is the first hospital system globally to participate in a landmark study using a first-of-its-kind EEG device.
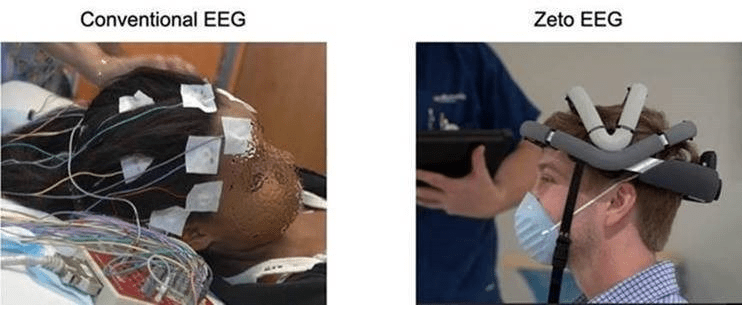
A press release states that EEG (electroencephalogram) tests are conducted to detect problems in the brain that may be associated with certain brain disorders such as seizures, tumors or strokes. A typical EEG test consists of multiple electrodes attached to a patient’s scalp with adhesive. Those electrodes have several wires connected to an amplifier which further connects to a computer that measures and records the results.
The setup and removal of a typical EEG test takes between 20–30 minutes. This new US Food and Drug Administration (FDA)-approved product, called zEEG from Zeto, (headquartered in Santa Clara, USA), can be set up in less than five minutes; which offers a more efficient and comfortable experience for the patient.
The patient wears equipment that looks like a high-tech bicycle helmet. The electrodes do not need to be glued to the patient’s scalp, do not leave any residue on the patient and there are no wires connected to an amplifier or computer.
An EEG technician at Methodist University can now be more productive and perform more EEGs in a day. The EEG and video recording are uploaded to the cloud where a specialist can read them at their own convenience.
The ZETO Instant EEG has been beneficial in the midst of the unprecedented COVID-19 pandemic. Due to the convenience of this EEG technology, personal protective equipment (PPE) and exposure time for healthcare workers is reduced significantly. Fast setup and clean up decrease the exposure times for nurses and other care providers. The ZETO Instant EEG uses single-use electrodes which also reduces the risk of contamination.










