The final results of the ESCAPE-NA1 trial have been presented and published contemporaneously in The Lancet. The study investigated the safety and efficacy of nerinetide—a neuroprotectant drug—in subjects undergoing endovascular thrombectomy. At the International Stroke Conference (ISC; 19–21 February, Los Angeles, USA), Michael Hill and Mayank Goyal, University of Calgary, Calgary, Canada, concluded that nerinetide was not superior to placebo. However, effect modification by alteplase was observed.
“When all patients were included; patients who received both alteplase and no alteplase, there was only a small effect size [2.1%],” explained Hill, adding that this demonstrates non-significance between modified Rankin Scale (mRS 0–2) scores at 90-days in nerinetide and placebo patients (61.3% vs. 59.2%, respectively).
Yet, evidence of a treatment effect modification was present, resulting in inhibition of treatment effect in patients receiving alteplase. Discussing the interaction, Hill said that in around 40% of patients who did not receive the thrombolytic, nerinetide was associated with improved functional outcomes, a lower risk of mortality, and reduced infarct volume. “The overall effect size is quite large: 9.5% absolute risk difference [in mRS scores at 90-days]; the number needed to treat [NNT] around 11 patients, with an adjusted risk ratio of 1.18.
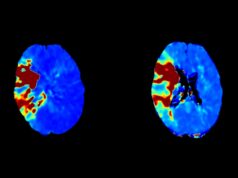
“We also, remarkably, see a mortality reduction,” Goyal added, alluding to the 7.5% absolute mortality benefit in the no alteplase stratum that received nerinetide. The latter finding corresponded with a reduction in infarct volumes (39.2 vs. 26.7mL reduction in the nerinetide and placebo group, respectively), which were measured blinded to outcome, he said.
They speculated that the treatment effect appears to come about because of a “substantial” reduction in the available nerinetide in patients receiving alteplase. “We have a small group of patients from which we got pharmacokinetic data. We think that there is clear biological evidence that the reason we saw no effect in the alteplase group was that the alteplase was activating proteases, which were chewing up the drug.”
It was these interactions that led Hill to conclude: “Neuroprotection in humans is possible”, as he asserted that further studies of nerinetide are needed to explore its effects.
In relation to the safety results, Hill reported that an equivalent number of serious adverse events occurred in each group: 35.7% in the placebo arm, with 33.1% for nerinetide patients.
Discussing the rationale behind the study of neuroprotection in humans, Goyal addressed delegates at the ISC, and said: “Over 1,000 compounds have been tested for neuroprotection and around 10% of those have been tested in humans. But as of right now, no neuroprotectants have shown clinical benefit.
“Nerinetide, or NA-1, is a promising agent that has shown neuroprotection in cell cultures, rodents, primates, and in a phase 2 study—the ENACT study,” he added. Published in 2012 in The Lancet Neurology, ENACT included patients undergoing endovascular treatment for aneurysms. According to Goyal, it demonstrated the safety of nerinetide and showed a reduction in the degree of diffusion hits and volume in patients treated with nerinetide.
Prior work in primates was carried out using an ischaemic-reperfusion model. “A middle cerebral artery occlusion was produced, and after waiting for a certain period of time, the drug was given. After 30 minutes the clip was removed to maintain reperfusion. This was able to show a significant reduction in infarct volume in the primates who got NA-1, but more importantly, there was a significant clinical improvement as measured by the National Institutes of Health Primate Stroke Scale (NIHPSS), where the primates who received the drug performed better,” explained Goyal.
ESCAPE-NA1 aimed to recapitulate the primate model in community-onset ischaemic stroke, accounting for existing standards of care including alteplase. It was a phase 3, multicentre, blinded, placebo-controlled, parallel group, single-dose design.
“We had the intention of enrolling 1,120 subjects, randomised 1:1 to nerinetide to placebo, stratified by alteplase use and by declared first choice of device,” said Goyal. All the patients had proximal vessel occlusion and underwent endovascular therapy, while ASPECTS ≥5 were allowed to be enrolled.
Goyal acknowledged that the inclusion criteria were “quite standard”. The five aspects he highlighted included: acute ischaemic stroke suitable for immediate endovascular treatment, age 18 or older, an onset time to randomisation within 12 hours, a baseline NIHSS greater than 5 at the time of randomisation, and pre-stroke independent functional status in activities of daily living with modified Barthel Index (BI) >90 (95 or 100). “The imaging criteria was similar to what was used in the ESCAPE trial, we just made it slightly more lenient as an ASPECTS 5 was allowed to be included.
“We used multiphase CT angiography to determine collaterals, and patients with moderate to good collaterals were necessary for the patient to be included. Part of our thinking was that the collaterals were necessary to allow the study drug to reach the ischaemic issue,” noted Goyal.
Alluding to details of the intervention, Hill said it involved a single, 10-minute infusion of nerinetide. The weighted dose was 2.6mg/kg, which was administered “as soon as possible” in the whole process of treating the patient. “We had a target of randomisation to study drug infusion time within 30 minutes, which we achieved with only a median time of 11 minutes,” he confirmed.
According to Hill, ESCAPE-NA1 had 48 sites around the world, primarily in Canada and the USA. The investigators stopped the trial once 1,105 patients were enrolled, which took under just two years.