
On technological and commercial fronts alike, the neuromodulation space is one of the more rapidly evolving subsectors within the medical industry—and has undergone a remarkable period of growth in the past couple of decades. With this progress showing little sign of slowing any time soon, Marc Russo (Hunter Pain Specialists, Newcastle, Australia) assesses the current landscape in brain stimulation therapies, and provides a glimpse of what the future may yet hold.
When I think back over the history of neuromodulation, I marvel at several things. Firstly, how quickly theory became reality after the Gate Control Theory of Pain was published and Norman Shealy’s pioneering first implant.1,2 Secondly, how slowly things moved for a generation of physicians as incremental progress was made on adding contacts, shrinking batteries, decreasing device failures, and identifying responder patients. Thirdly, how progress has exploded over the last 15 years with the arrival of multiple new neuromodulation companies—spearheaded by Nevro Corporation, founded by Konstantinos Alataris in 2007—as well as sophisticated models of neural transmission that have informed waveform development. This process is continuing to play out.
As of 2022, we have companies in this space with multiple waveform patterns, waveforms to stimulate neurones and glial cells in concert, energy-efficient waveforms that allow primary cell pulse generators, closed-loop monitoring of spinal cord electrical compound action potentials, ultra-low frequency waveforms, ultra-high frequency waveforms, and battery-less pulse generators.3 There is not space to elaborate on all the potential and the deliverables that they offer patients.
The common theme from the above is a desire to understand the neurophysiology of pain, of spasticity, of gait disorders and of motor weakness, and to harness that knowledge into building waveforms and devices that efficiently and successfully interact with the nervous system to inhibit or augment nerve signalling to achieve the clinical ends. Where once we forced the nervous system to respond to tonic (repetitive and unvarying) square waves, because it was easy to see on an oscilloscope and easy to build into a pulse generator, we now accept and embrace complexity to achieve our targets.
In spinal cord stimulation (SCS) and deep brain stimulation, we are seeing complex waveforms better interface with the nervous system at the same time as the patient’s experience with their device interaction becomes much more seamless and naturally intuitive. I am glad that Apple and Google train so many engineers, because some of them end up in the neuromodulation space and bring their concepts of ‘lifestyle experience’ into the design ecosystem.
A second major development in this field is understanding where we are helping or hindering our patients by their adjuvant treatment. Nissen et al have shown that opioids (morphine >40mg/day) increase the explant rate of SCS, while drugs that decrease central sensitisation (i.e., augment descending inhibition) decrease explant rates of SCS.4 The figure can swing from worst-case 30% explants to best-case 0% explants depending on just these two factors. I think it likely that understanding how the endocannabinoid system interfaces with neuromodulation devices may refine this further.
We are also in the ongoing process of moving neuromodulation from its traditional cottage industry of relatively isolated practitioners doing low-volume implants to integrated high-volume centres of excellence with evaluation, implant, holistic optimisation, and functional rehabilitation, audit, research and publishing. This will yield improvements as big as (or more so) addressing the technology shortfalls. The International Neuromodulation Society is very much engaged in this area through both Neurostimulation Appropriateness Consensus Committee (NACC) and Polyanalgesic Consensus Committee (PACC) articles, and Centers of Excellence development programme.5
Lastly, the elephant in the room for which we hold much promise in—machine learning or artificial intelligence being designed into devices. Independent thinking software running the hardware (within defined limits) combined with input from the patient (in a bewildering variety of inputs, some non-intuitive) as well as with meta data analysis of thousands/millions of patient inputs and outputs held in the cloud; where will these lead to? That is hard to say, but the devices I cut my teeth on in the 1990s will be seen as the electronic version of rubbing two sticks together.
I have both high hopes for and great faith in what is to come for neuromodulation as we traverse the 2020s. Devices are improving at something beyond a linear rate. Medical provider and patient awareness of therapy is improving, albeit not as fast as we would like. Third party payors are gradually seeing the cost-effectiveness data come through that they need to see to support the therapy (why did the field ignore this aspect for so long, I wonder?), and governmental and regulatory bodies are seeing the unique solutions that this therapy can provide across multiple refractory conditions.
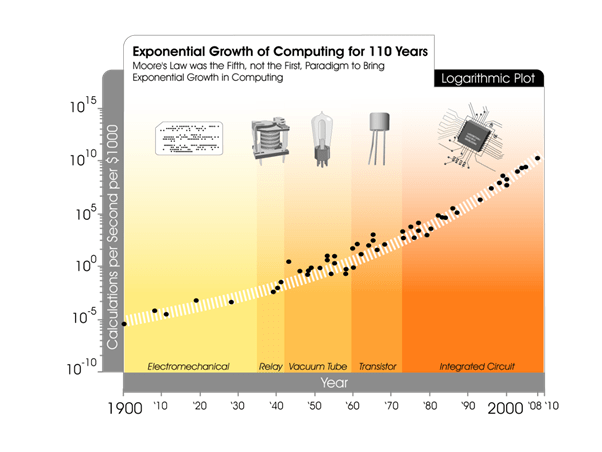
Is there anything here that is the odd man out? Well, Moore’s Law reinterpreted by Ray Kurzweil states that cost should come down as computing power goes up over time.6 I think to a certain extent neuromodulation is a bit of a holdout in regards to that and I predict that, over time, we will see it follow Kurzweil’s graph (Figure 1) and cost reduction will be the next frontier to be crossed in our area.
References:
- Melzack R, Wall P D. Pain mechanisms: a new theory. Science. 1965; 150(3699):971-9.
- Shealy C N, Mortimer J T, Reswick J B. Electrical Inhibition of Pain by Stimulation of the Dorsal Columns. Anesthesia & Analgesia. 1967; 46(4):489-91.
- Provenzano D A, Heller J A, Hanes M C. Current Perspectives on Neurostimulation for the Management of Chronic Low Back Pain: A Narrative Review. Journal of Pain Research. 2021; 14:463-479.
- Nissen M, Ikäheimo T M, Huttunen J, et al. Gabapentinoids Associated With Lower Explantation Rate in 203 Patients With Spinal Cord Stimulation for Failed Back Surgery Syndrome. Neurosurgery. 2021; 89(4):626-634.
- International Neuromodulation Society Committees. https://www.neuromodulation.com/ins-committees. Accessed 7 February 2022.
- Kurzweil R. The age of spiritual machines: When computers exceed human intelligence. New York, Viking, 1999.
Marc Russo is the director of Hunter Pain Specialists in Newcastle, Australia and current president of the International Neuromodulation Society (INS).









