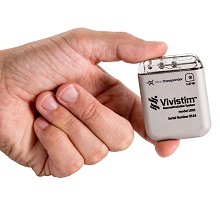
MicroTransponder today announced that its Vivistim paired vagus nerve stimulation (VNS) system has won the Neurotech Reports Gold Electrode Award for best new product. This announcement comes as MicroTransponder prepares for the first commercial implantation of its Vivistim system and as the company plans a US sales launch next year.
The US Food and Drug Administration (FDA)-approved Vivistim system employs paired VNS that is activated during rehabilitation therapy to improve upper limb function for stroke survivors. Paired VNS therapy generates two–to–three times more hand and arm function than intense rehabilitation alone after six weeks of in-clinic therapy, a MicroTransponder press release states.
“Our editors selected MicroTransponder’s Vivistim system as the best new product of 2021 because it breaks new ground in stroke neurorehabilitation, and helps open the door for funding and commercialisation of new neurorehabilitation systems,” said James Cavuoto, editor and publisher of Neurotech Reports.
The Vivistim system received FDA premarket approval in August 2021, following a decade of preclinical research and a successful 108-person, multicentre, double-blinded, randomised controlled trial that was peer-reviewed and published in The Lancet. MicroTransponder has also applied for a CE mark for the Vivistim system.
“We are honoured that Neurotech Reports recognised Vivistim as a breakthrough neurotechnology that addresses an unmet need for stroke survivors and their healthcare providers,” said Jordan Curnes, co-founder, president and CEO of MicroTransponder. “As we begin commercialisation of Vivistim, we remain committed to helping stroke survivors achieve improvements in their functional mobility while enhancing their quality of life. We are also proud to join the ranks of previous Neurotech Reports award winners including Axonics and Saluda Medical.”













