Tag: Senza II
Nevro receives TGA approval for Senza II spinal cord stimulation system...
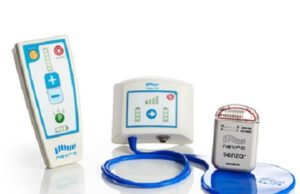
The Therapeutic Goods Administration (TGA) has granted approval to Nevro for its next-generation Senza II spinal cord stimulation system delivering the company’s proprietary HF10...
FDA approves Senza II Spinal Cord Stimulation System
The US Food and Drug Administration has approved the next-generation Senza II Spinal Cord Stimulation (SCS) System (Nevro) delivering HF10 therapy.
According to the company,...