 Oxford Heartbeat has shared that the clinical impact trial of its decision-support software, PreSize Neurovascular, has been endorsed by the European Society of Minimally Invasive Neurological Therapy (ESMINT).
Oxford Heartbeat has shared that the clinical impact trial of its decision-support software, PreSize Neurovascular, has been endorsed by the European Society of Minimally Invasive Neurological Therapy (ESMINT).
As stated in an Oxford Heartbeat press release, ESMINT endorsement “signifies strong support from a leading authority in neurointerventional medicine”, and reinforces the company’s mission to “set a new standard of evidence generation for the use of decision-support software in neuroradiology”.
The prospective, multicentre trial—which aims to quantify the benefits of using PreSize in clinical practice—is being independently run by Imperial Clinical Trials Unit (ICTU) in London, UK. The trial involves nine UK hospitals and 22 interventional neuroradiologists, and successfully completed recruitment of 103 cerebral aneurysm patients last year. It has been funded by the UK National Institute for Health and Care Research’s (NIHR) AI in Health and Care Award, and is described by Oxford Heartbeat as “the first trial of its kind” for decision-support software in the neurovascular space.
“ESMINT plays a crucial role in shaping clinical practice in neurointervention, so [its] endorsement of the PreSize trial highlights the trial’s robust methodology, and validates its scientific and clinical relevance,” said Tufail Patankar (Havering and Redbridge University Hospitals NHS Trust, London, UK), the trial’s chief investigator.
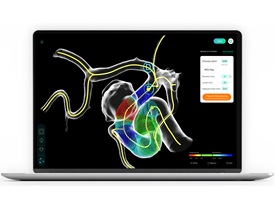
The 103 patients included in the study were diagnosed with a cerebral aneurysm and scheduled for minimally invasive treatment. For each patient, treatment was planned twice—both with and without PreSize—and information was collected on the accuracy of the software as well as surgical efficiency metrics and patient outcomes.
The results of the trial will be used to compare clinical decision-making with traditional planning methods versus with PreSize, with data currently undergoing independent analysis by ICTU. Publication of these results is anticipated later this year, according to Oxford Heartbeat.
“This trial is so exciting because it is the first in the field where the data are collected in a real-world, prospective setting to demonstrate objective and measurable benefits that PreSize brings to patients and clinical teams,” stated Katerina Spranger, chief executive officer (CEO) and founder of Oxford Heartbeat.
The company further claims in its recent release that publication of the trial results will position PreSize as “the only software of its kind” to be supported by Level 1 evidence, as per American Journal of Neuroradiology guidelines.
“ESMINT endorsement will increase the visibility of the trial across the European neurovascular community, opening the door for wider collaboration, support, and adoption of the technology in clinical practice,” Patankar added.









