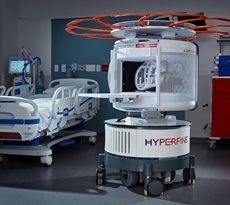
Hyperfine has announced receipt of CE marking and UK Conformity Assessment (UKCA) approval for both the next-generation Swoop system and the latest advancement in its Optive AI software. The company notes that, together, these regulatory approvals enable commercialisation of Hyperfine’s most advanced portable magnetic resonance imaging (MRI) technology across Europe and the UK, expanding access to high-quality brain imaging at the point of care and representing “a pivotal milestone” in its international commercial growth strategy.
The next-generation Swoop scanner—which was cleared by the US Food and Drug Administration (FDA) in 2025—represents a “significant advancement” in portable brain MRI, delivering “substantial improvements” in image quality, workflow, and overall user experience. The next-generation Swoop system has generated “strong customer interest” in the US market, where Hyperfine has placed systems in critical care and emergency rooms of hospitals as well as in neurology offices, according to the company.
In addition, Hyperfine has received European approvals for the latest advancement in its Optive AI software, which was also cleared by the US FDA last year. The updated software includes a new multi-direction diffusion-weighted imaging (DWI) sequence that acquires and averages images from multiple diffusion directions—similarly to the method used in high-field MRI scanners. Hyperfine notes that this capability produces cleaner, more consistent images that are “especially valuable” for stroke diagnosis, enabling better detection of small strokes and enhancing the value of the Swoop system in acute neurological care.
The Optive AI software is part of the next-generation Swoop system and is also available as a standalone software upgrade, meaning existing users “can benefit from the same powerful imaging platform”, Hyperfine further claims.
“We are proud to bring our most innovative technology to clinicians and patients in Europe and the UK, and are thrilled to have achieved these approvals ahead of our internal expectations,” said Maria Sainz, president and chief executive officer (CEO) of Hyperfine. “This milestone is a testament to the exceptional execution of our product and regulatory teams, and meaningfully advances a key pillar of Hyperfine’s 2026 international growth strategy. We believe that the next-generation Swoop system will accelerate international adoption of our technology and strengthen our position as the global leader in AI [artificial intelligence]-powered portable brain imaging.”
Hyperfine plans to initiate commercialisation of the next-generation Swoop system and the latest Optive AI software, both in Europe and the UK, early in the third quarter of 2026.









